Class 10 Science MCQ Chapter 2 Acids, Bases and Salts, Class 10 Science MCQ Question Answer, Class 10 Science Multiple Choice Question Answer to each chapter is provided in the list of SCERT Science Class 10 Objective Types Question Answer so that you can easily browse through different chapters and select needs one. Class 10 Science MCQ Chapter 2 Acids, Bases and Salts Question Answer can be of great value to excel in the examination.
SCERT Class 10 Science MCQ Chapter 2 Acids, Bases and Salts
SCERT Class 10 General Science MCQ Chapter 2 Acids, Bases and Salts Notes covers all the exercise questions in SCERT Science Textbooks. The NCERT Class 10 Science MCQ Chapter 2 Acids, Bases and Salts provided here ensures a smooth and easy understanding of all the concepts. Understand the concepts behind every chapter and score well in the board exams.
Acids, Bases and Salts
Chapter – 2
Multiple Choice Questions & Answers
1. Human body works efficiently within the pH range
(a) Lower than 5.6
(b) Lower than 5.5
(c) Between 7.0 and 7.8
(d) Between 0 and 14
Ans: (c) Between 7.0 and 7.8
2. Our tooth decay starts when pH of mouth is-
(a) Less than 7.0
(b) Less than 5.5
(c) More than 5.6
(d) More than 5.5
Ans : (b) less than 5.5
3. Which of the following acid is secreted when nettle leaves sting-
(a) Acetic acid.
(b) Citric acid.
(c) Lactic acid.
(d) Methanoic acid.
Ans: (d) Methanoic acid.
4. The nature of sodium chloride salt-
(a) Neutral.
(b) Basic.
(c) Acidic.
(d) None of these.
Ans: (a) Neutral.
5. The nature of salt produced by the reaction between a strong and a weak base-
(a) Basic.
(b) Acidic.
(c) Neutral.
(d) None of these.
Ans: (b) Acidic.
6. The nature of salt produced by the reaction between a weak acid and a strong base-
(a) Basic.
(b) Neutral.
(c) Acidic.
(d) None of these.
Ans: (a) Basic.
7. Which of the following non-metallic oxides is neutral in nature-
(a) Neutral.
(b) Acidic.
(c) Basic.
(d) None of these.
Ans: (a) Neutral.
8. Which of the following non-metallic oxides is neutral in nature –
(a) CO₂
(b) SO₂
(c) H₂O
(d) SO₃
And: (c) H₂O
9. Which of the following sodium compounds is a component of antacids-
(a) Sodium hydroxide.
(b) Sodium hydrogen carbonate.
(c) Sodium carbonate.
(d) Bleaching powder.
Ans: (b) Sodium hydrogen carbonate.
10. Which of the following is used to remove permanent hardness of water-
(a) Washing soda.
(b) Baking soda.
(c) Sodium hydroxide.
(d) Bleaching powder.
Ans: (a) Washing soda.
11. Which of the following compounds produces bleaching powder by the action of chlorine-
(a) Sodium hydroxide.
(b) Calcium hydroxide.
(c) Sodium carbonate.
(d) Potassium hydroxide.
Ans: (b) Calcium hydroxide.
12. The compound formed when Plaster of Paris reacts with water is-
(a) Copper sulphate.
(b) Sodium carbonate.
(c) Baking Soda.
(d) Gypsum.
Ans: (d) Gypsum.
13. A solution has turned blue litmus paper red. Its pH is likely to be-
(a) 1
(b) 7
(c) 8
(d) 10
Ans: (a) 1
14. Which of the following is heated to obtain sodium carbonate-
(a) Slaked lime.
(b) Baking soda.
(c) Gypsum.
(d) Plaster of Paris.
Ans: (b) Baking soda.
15. Chlor-alkali process gives-
(a) Sodium hydroxide.
(b) Chlorine.
(c) Hydrogen.
(d) All of above.
Ans: (d) All of above.
Very Short & Short Type Questions and Answers:
1. What are indicators?
Ans: The substances which change colour in acidic and basic solutions to help in the identification of acids and bases are called indicators.
2. Give example of two natural indicators.
Ans: Litmus, turmeric.
3. Give example of two synthetic indicators?
Ans: Methyl orange, phenolphthalein.
4. What are olfactory indicators?
Ans: The substances which change odour in acidic and basic solutions are called olfactory indicators.
5. Give three examples of olfactory indicators.
Ans: Onion, vanilla clove.
6. What is universal indicator?
Ans: It is a mixture of several indicators which shows different colours at different concentration of hydrogen ions in a solution.
7. Which gas is generally liberated when acids react with a metal?
Ans: Hydrogen.
8. Which gas is liberated when acids react with metal carbonates and hydrogen carbonates?
Ans: Carbon dioxide.
9. What are the products of the reaction between an acid and a base?
Ans : Salt and water.
10. What is the hydronium or oxonium ion?
Ans: The H⁺ ion produced by an acid in aqueous solution immediately reacts with water to get converted into H₃O⁺ ion. This is known as hydronium or oxonium ion.
11. What do you mean by dilution of acids and bases?
Ans : Mixing an acid or base with water results in decrease in the concentration of ions (H₃O⁺ / OH⁻) per unit volume. Such a process is called dilution.
12. What is pH scale?
Ans: A scale which is used to measure hydrogen ion concentration in a solution is called pH scale.
13. What is the range of pH scale?
Ans: From 0 to 14.
14. What is acid rain?
Ans: When the pH of rain water is less than 5.6, it is called acid rain.
15. What is chlor-alkali method?
Ans: When electricity is passed through an aqueous solution of sodium chloride (called brine), it decomposes to form sodium hydroxide, chlorine and hydrogen. This is called chlor-alkali method.
16. Name the sodium compound produced by chlor-alkali process.
Ans: Sodium hydroxide.
17. What is brine?
Ans: An aqueous solution of sodium chloride is called brine.
18. Write the chemical formula of bleaching powder.
Ans: CaOCl₂
19. What is the chemical of backing soda?
Ans: Sodium hydrogen carbonate.
20. What is backing powder?
Ans: Baking powder is a mixture of baking soda and a mild, edible acid such as tartaric acid.
21. Write the chemical formula of washing soda.
Ans: Na₂CO₃. 10H₂O
22. What is the chemical name of washing soda?
Ans: Hydrated sodium carbonate.
23. What is water of crystallisation?
Ans: Water of crystallisation is the fixed number of water molecules present in one formula unit of a salt.
24. Give example of two hydrated salt containing water of crystallisation.
Ans: Copper sulphate (CuSO₄. 5H₂O), Gypsum (CaSO₄.2H₂O).
25. What is plaster of Paris?
Ans: It is a white powder which is chemically known as calcium sulphate hemihydrate.
CaSO₄.1/2H₂O
26. Only half a water molecule is shown to be attached as water of crystallisation in a formula unit of plaster of Paris. Why?
Ans: This is because, two formula units of CaSO₄ share one molecule of water in Plaster of Paris.
27. How is Plaster of Paris Produced?
Ans: On heating gypsum at a 373 K, it loses water molecules and gets converted into Plaster of Paris.
28. Which salt is produced when hydrochloric acid reacts with sodium hydroxide? Write the reaction.
Ans: Sodium chloride;
HCl + NaOH → NaCl + H₂O
29. What is the nature of sodium chloride? What is its pH value?
Ans: Neutral. Its pH is 7
30. What is bleaching powder?
Ans: It is a white powder which is chemically known as calcium oxychloride (CaOCl₂).
31. What is baking soda?
Ans : It is a mild, noncorrosive basic salt chemically known as sodium hydrogen carbonate. [NaHCO₃]
32. What is washing soda?
Ans: It is a basic salt chemically known as hydrated sodium carbonate. [Na₂CO₃.10H₂O]
33. What is neutralisation reaction?
Ans: When an acid reacts with a base, salt and water are produced. This reaction is called neutralisation reaction.
34. Give an example of a neutralisation reaction.
Ans : HCl + NaOH → NaCl + H₂O
35. Phenol -phthalein turns pink in a solution. What is the nature and pH of the solution?
Ans : Nature of solution: Basic; pH: more than 7.
36. What are produced when an acid reacts with a metal?
Ans: A salt and hydrogen gas.
37. Give example of between an acid and a metal.
Ans: Zn + H₂SO₄ → ZnSO₄ + H₂.
38. What happens when a acid reacts with metal carbonate or metal hydrogen carbonate?
Ans: A salt, carbon dioxide and water are produced.
39. Give an example of a reaction between an acid and metal carbonate.
Ans : 2HC l+ Na₂CO₃ → 2NaCl + CO₂ + H₂O
40. Give an example of a reaction between an acid and a metal hydrogen carbonate.
Ans : HCl + NaHCO₃ → 2NaCl + CO₂ + H₂O
41. The lime water turns milky when carbon dioxide is passed through it. Why?
Ans: Due to the formation of a white precipitate called calcium carbonate in the. reaction between CO₂ and lime water.
42. Write the chemical equation for the reaction between CO₂ and lime water.
Ans: Ca (OH)₂ + CO₂ → CaCO₃ + H₂O
43. Why does milky lime water turn clear again on passing excess CO₂ through it?
Ans: Due to formation of calcium hydrogen carbonate which is soluble in water.
44. Write the reaction that takes place when excess carbon dioxide is passed through milky lime water.
Ans : CaCO₃ + H₂O + CO₂ → Ca(HCO₃)₂
45. How is the reaction between an acid and a base known as?
Ans: Neutralisation réaction.
46. What are produced when an acid reacts with a metal oxide?
Ans: A salt and water.
47. Give an example of a reaction between an acid and a metal oxide.
Ans: 2HCl + CuO → CuCl₂ + H₂O
48. If you add some dilute hydrochloric acid in copper oxide taken in a beaker, what will be the colour of the solution?
Ans: Blue-green.
49. If you add dilute hydrochloric acid to copper oxide taken in a beaker, what will happen to the copper oxide?
Ans: The copper oxide will dissolve.
50. What is the nature of metallic oxides?
Ans: Basic.
51. What are produced when a base reacts with a metal?
Ans: A salt and hydrogen gas.
52. Give an example of a reaction between a base and a metal.
Ans: 2NaOH + Zn→ Na₂ ZnO₂ + H₂
53. Place a few pieces of granulated zinc metal in a test tube and add 2mL of sodium hydroxide solution to them. Write a balanced chemical equation for the reaction occurred.
Ans: 2NaOH + Zn→ Na₂ ZnO₂ + H₂
54. What are produced when a non-metallic oxide reacts with base?
Ans: A salt and water.
55. What is the nature of non-metallic oxides?
Ans: Acidic.
56. Give an example of a reaction between a non metallic oxide and a base.
Ans: Ca (OH)₂ + CO₂ → CaCO₃ + H₂O
57. Name the sodium compound produced when electric current is passed through Brine.
Ans: Sodium hydroxide (NaOH)
58. Which substances are produced when electric current is passed through Brine?
Ans: Sodium hydroxide, chlorine and hydrogen.
59. Write the chemical reaction that takes place when electric current is passed through Brine.
Ans:
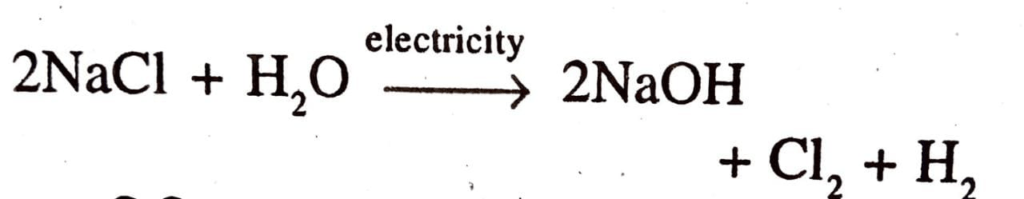
60. Write a balanced chemical equation for the reaction occurred during the preparation of bleaching powder.
Ans: Ca(OH)₂ + Cl₂ → CaOH₂ + H₂O (bleaching powder).
61. How is bleaching powder prepared?
Ans: By the action of chlorine on dry slaked lime.
62. How is baking soda prepared?
Ans: By the reaction between sodium chloride, water, carbon dioxide and ammonia.
63. Write the chemical reaction used to produce baking soda?
Ans: NaCl + H₂O + CO₂ + NH₃ → NH₄Cl + NaHCO₃ (Baking soda)
64. Write the reaction that takes place when baking powder is heated or mixed in water.
Ans: NaHCO₃ + H⁺ → CO₂ + H₂O + (from acid) + Sodium salt
65. What will happen if a sodium hydrogen carbonate is heated?
Ans: Sodium carbonate, carbon dioxide and water will be produced.
66. Write the reaction that takes place when sodium hydrogen carbonate (baking soda) is heated?
Ans:
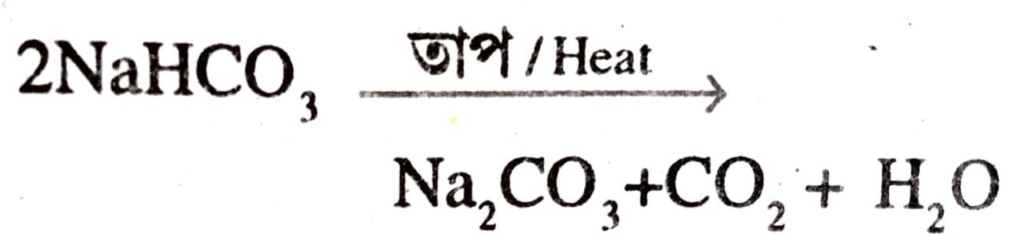
67. Heat a few crystals of copper sulphate in a dry boiling tube. What will be the colour of copper sulphate after heating?
Ans: White.
68. Take about 0.5g of sodium carbonate in a test tube and take about 0.5g of sodium hydrocarbonate in another test tube. Add 2mL of dilute HCI in both the test tubes. What will you observe?
Ans: Bubbles of CO₂ gas will be observed in both the test tubes.
69. Take some granules of zinc in a test tube and add a solution of sodium hydroxide to them and warm the test tube. What will you observe?
Ans: Bubbles of hydrogen gas will be observed to evolve.
70. Take about 1g solid NaCl in a clean and dry test tube and add some dilute sulphuric acid to the test tube. A gas will be observed coming out of the delivery tube. Identify the gas.
Ans: Hydrogen chloride (HCl).
71. Take about 2mL dilute NaOH solution in a test tube and add two drops of phenolphthalein solution. What will be the colour of the solution?
Ans: Pink.

Hi! my Name is Parimal Roy. I have completed my Bachelor’s degree in Philosophy (B.A.) from Silapathar General College. Currently, I am working as an HR Manager at Dev Library. It is a website that provides study materials for students from Class 3 to 12, including SCERT and NCERT notes. It also offers resources for BA, B.Com, B.Sc, and Computer Science, along with postgraduate notes. Besides study materials, the website has novels, eBooks, health and finance articles, biographies, quotes, and more.




