Class 11 Biology Chapter 9 Biomolecules, AHSEC Class 11 Biology Question Answer, HS 1st year Biology notes to each chapter are provided in the list so that you can easily browse throughout different chapters Assam Board Class 11 Biology Chapter 9 Biomolecules Question Answer and select needs one.
Class 11 Biology Chapter 9 Biomolecules
Also, you can read the SCERT book online in these sections Solutions by Expert Teachers as per SCERT (CBSE) Book guidelines. These solutions are part of SCERT All Subject Solutions. Here we have given Assam Board Class 11 Biology Chapter 9 Biomolecules Solutions for All Subjects, You can practice these here.
Biomolecules
Chapter – 9
VERY SHORT ANSWER TYPE QUESTIONS
Q.1. What are biomolecules?
Ans :- All the carbon compounds that we get from living tissues are called biomolecules.
Q.2. From biological point of view, classify the organic or inorganic compound?
Ans :- From a biological point of view, compounds are classified into amino acid, nucleotide base, fatty acid etc.
Q.3. What are amino acids?
Ans :- Amino acid are organic compound containing an amino group and an acidic group as substituents on the same carbon ie x-carbon.
Q.4. What is a particular property of amino acid ?
Ans :- A particular property of amino acid is the ionizable nature of – NH2 and COOH group.
Q.5. How many carbon atoms are present in palmitic acid and arachidonic acid ?
Ans :- There are 16 carbon atom in palmitic acid and 20 carbon atoms in arachidonic acid.
Q.6. What are fatty acids ?
Ans :- Fatty acids are organic compounds made up of long hydrocarbon chain which contain a terminal carboxyl group (-COOH) e.g acetic acid (CH3 COOH).
Q.7. What are fats ?
Ans :- Fats are the esters of fatty acid with glycerol.
Q.8. What is oil ?
Ans :- Oil are group of simple lipid rich in unsaturated fatty acid and remain liquid at room temperature.
Q.9. What are phospholipids ?
Ans :- Some lipids have phosphorus and a phosphory lated organic compound in them. These are called phospholipids.
Q.10. Write the name of two phospholipid ?
Ans :- Lecithin and cephalin are phospholipid.
Q.11. Define nucleoside.
Ans :- A nucleoside is the product of condensation of the nitrogenous base and a pentose sugar.
Q.12. Define nucleotide.
Ans :- A nucleotide is the product of condensation of a nucleoside and a phosphoric acid molecule.
Q.13. Give examples of nucleoside.
Ans :- Adenosine, guanosine, thymidine, uridine and cytidine are nucleotides.
Q.14. Give example of nucleotide .
Ans :- Adenylic acid, thymidine acid, guanylic acid uridylic acid and cytidylic acid are nucleotides.
Q.15. What are primary metabolites ?
Ans :- The organic compounds including amino acid, sugar, and lipids etc are called primary metabolites.
Q.16. What type of organic compound are present in t eh acid insoluble fraction ?
Ans :- The organic compound present in the acid insoluble fraction are protein, nucleic acid, polysaccharides.
Q.17. What are macromolecules ?
Ans :- The chemical compounds which have molecular weight less than one thousand dalton and are referred to an macromolecule.
Q.18. What are proteins ?
Ans :- Proteins are linear chains of amino acid linked by peptide bond.
Q.19. Which is the most abundant protein in animal world and which is the most abundant protean in the whole of biosphere.
Ans :- Collagen is the most abundant protein in animal world and Ribulose biphosphate carboxylase Oxygenase (RUBISCO) is the most abundant protein in the whole of biosphere.
Q.20. What are polysaccharide ?
Ans :- Polysaccharide are long chains of sugars e.g. cellulose.
Q.21. Give three examples where cellulose is present.
Ans :- Cellulose is present in plants cell wall, plant pulp and cotton fibre.
Q.22. Where is chitin found ?
Ans :- Exoskeleton of arthropods have a complex polysaccharide called chitin.
Q.23. What are the distinct components of nucleotide ?
Ans :- A nucleotide has three chemically distinct components one is a heterocyclic compound, the second is a monosaccharide and the third is a phosphoric acid or phosphate.
Q.24. What is purine and pyrimidine ?
Ans :- Nucleic acid contains nitrogenous bases named adenine, guanine, uracil, cytosine and thymine. Adenine and Guanine are purine while the rest are pyrimidines.
Q.25. What is the primary structure of a protein ?
Ans :- The sequence of amino acids i.e the positional information in a protein which is the first amino acid, which is second and so on is called the primary structure of a protein.
Q.26. What type of bond is present in polypeptides ?
Ans :- Amino acid the linked by peptide bond in a polypeptide or protein.
Q.27. Define an ester bond.
Ans :- The bond between the phosphate and hydroxyl group of sugar is called on ester bond.
Q.28. Who proposed the DNA (double helix) model ?
Ans :- Watson crick proposed the DNA model.
Q.29. What are enzymes ?
Ans :- The proteins with catalytic power are called enzymes.
Q.30. What are ribosomes ?
Ans :- When nucleic acid behave like enzymes, these are called ribosomes.
Q.31. What is the purpose of the ‘active site’ of enzyme ?
Ans :- The purpose of active site is to fit the substrate into it.
Q.32. Which enzymes retain their catalytic power even at high temperature ?
Ans :- Enzymes isolated from organism who normally live under extremely high temperature e.g hot vents and sulphur springs retain their catalytic power at high temperature.
Q.33. What is a physical change ?
Ans :- A physical change refers to a change in shape without breaking of bonds.
Q.34. Give an example of organic chemical reaction.
Ans :- Hydrolysis of starch into glucose is an example of organic chemical reaction.
Q.35. What is an inhibitor ?
Ans :- When the binding of the chemical shut off enzyme activity, the process is called inhibition and the chemical is called an inhibitor.
Q.36. What is competitive inhibitor ?
Ans :- When the inhibitor closely resemble the substrate in its molecular structure and inhibits the activity of the enzyme, it is known as competitive inhibitor.
Q.37. Define oxidoreductase enzyme.
Ans :- The enzyme which catalyze oxide reduction between two substrate e.g S and S 1.
S reduced + S¹ Oxidised + S¹ reduced
Q.38. What is lyase enzyme ?
Ans :- Enzyme that catalyse removal of groups from substrate by mechanism other than hydrolysis leaving double bonds.
X Y
1 1 ⇒ x – y + c = c
c – c
Q.39. What is transferase ?
Ans :- Enzymes catalysing a transfer of a group, G between a pair of substrate S and S¹ e.g.
S-D+S¹ → S+S¹-G
Q.40. What are coenzymes ?
Ans :- Coenzymes are organic compounds which serve as cofactors in a number of different enzyme catalysed reactions. e.g. NAD and NADP etc.
SHORT ANSWER TYPE QUESTIONS
Q.1. How does elemental analysis give elemental composition of living tissue ?
Ans :- When a living tissue is dried, all the water evaporates. Now if the tissue is fully burnt, all the carbon compounds are oxidised to gaseous form (Co2, water vapour) and are removed. The remains are called ash. This ash contains inorganic elements like calcium, magnesium etc.
So elemental analysis gives the elemental composition of living tissue.
Q.2. On what factor does the nature (Acid or base) of amino acid depend ?
Ans :- The acidic or basic nature of amino acid depends on the amino, carboxyl and R-functional groups.
Q.3. What are secondary metabolites ?
Ans :- When one analysis plant, fungal and microbial cells, one can see thousands of compounds other than these called primary metabolites e.g- alkaloids, rubber, essential oil, gums and spices. These are called secondary metabolites.
Q.4. What is the chief role of secondary metabolite ?
Ans :- Secondary metabolites are useful to human welfare e.g rubber, drugs, spices, sorts and pigments.
Q.5. What are the main functions of protein ?
Ans :- The main function of protein are –
(i) Protein carry transport nutrient across the cell membrane.
(ii) Protein fight against infectious organisms.
(iii) Some protein functions as hormones, enzymes.
Q.6. What is a phosphodiesterase bond ?
Ans :- In nucleic acid a phosphate moiety links the 3 – carbon of one sugar of one nucleotide to the 5 – carbon of one sugar of one nucleotide to the 5 carbon of the sugar of the succeeding nucleotide. The bond between the phosphate and hydroxide group of sugar is on ester bond. As there is one such ester bond on either side, it is called phosphodiesterase bond.
Q.7. All biomolecules have a turn outs what does this statement mean ?
Ans :- This means that the biomolecules are constantly being changed into some other biomolecules and also made from some other biomolecules. This breaking and making is through chemical reaction constantly occurring in living organism.
Q.8. Write a few examples of metabolic transformation.
Ans :- A few examples of metabolic transformation are removal of carbon dioxide from amino acids making an amino acid into a amine, removal of amino group in a nucleotide base, hydrolysis of a glycosidic bond in a disaccharide etc.
Q.9. Why is the chemical reaction considered catalysed reaction ?
Ans :- Every chemical reaction is a catalysed reaction. There is no uncatalysed metabolic conversation in living systems. Even Co2 dissolving in water, a physical process, is a catalysed reaction in living system. Be it a simple reaction or a complex one, each reaction needs a catalyst to proceed.
Q.10. What is the basic difference between anabolic and catabolic pathway ?
Ans :- The metabolic pathway by which complex structure are formed form simple structure are called anabolic pathway e.g. formation of cholesterol from acetic acid.
The metabolic pathway by which simple structure are formed from complex structure are called catabolic pathway. e.g- formation of lactic acid from glucose in skeletal muscle.
Anabolic pathway are called biosynthetic pathway and catabolic pathway are called degradation pathway.
Anabolic pathway consume energy whereas catabolic pathway release energy.
Q.11. Living state is a non-equilibrium steady state to be able to perform work comment.
Ans :- All living organism exist in a steady state characterised by concentration of biomolecular, The biomolecules are in a metabolic flux. The steady state is a non equilibrium stage. And when a system is at equilibrium, then no work is done. As living organism work continuously, they cannot afford to reach equilibrium.
So that living state is a non-equilibrium steady state to be able to perform work.
Q.12. Write the important properties of enzymes.
Ans :- The properties of enzyme are
i) Enzymes are biocatalyst. A small amount of enzyme can catalyst large amount of substrate.
ii) Enzymes are very specific in action.
iii) Enzymes are highly sensitive to heat (temperature).
iv) Enzymes are soluble in water, alcohol etc.
Q.13. What are the factors which influence the rate of reaction ?
Ans :- The factors which influence the rate of reaction are :
i) Temperature :- It is the most important factor which controls effectively the rate of reaction. The rate double or decreases by half for every 10°C change in either direction.
ii) Concentration of enzyme :- The rate of reaction increase with increase in concentration of enzyme.
e.g glucose becomes pyruvic acid in presence of many enzymes.
Q.14. What is activation energy ?
Ans :- When a pictorial representation is made having y-axis showing potential energy content and x-axis showing progress of reaction. Then two things are noticed. The energy level difference between S and P. If ‘P’ is at a lower level than ‘S’, the reaction is exothermal. Whether the reaction is exothermic or endothermic, the ‘S’ has to go through a much higher energy or transition state. The difference in average energy content of ‘S’ from that of this transition state is called, ‘activation energy’.
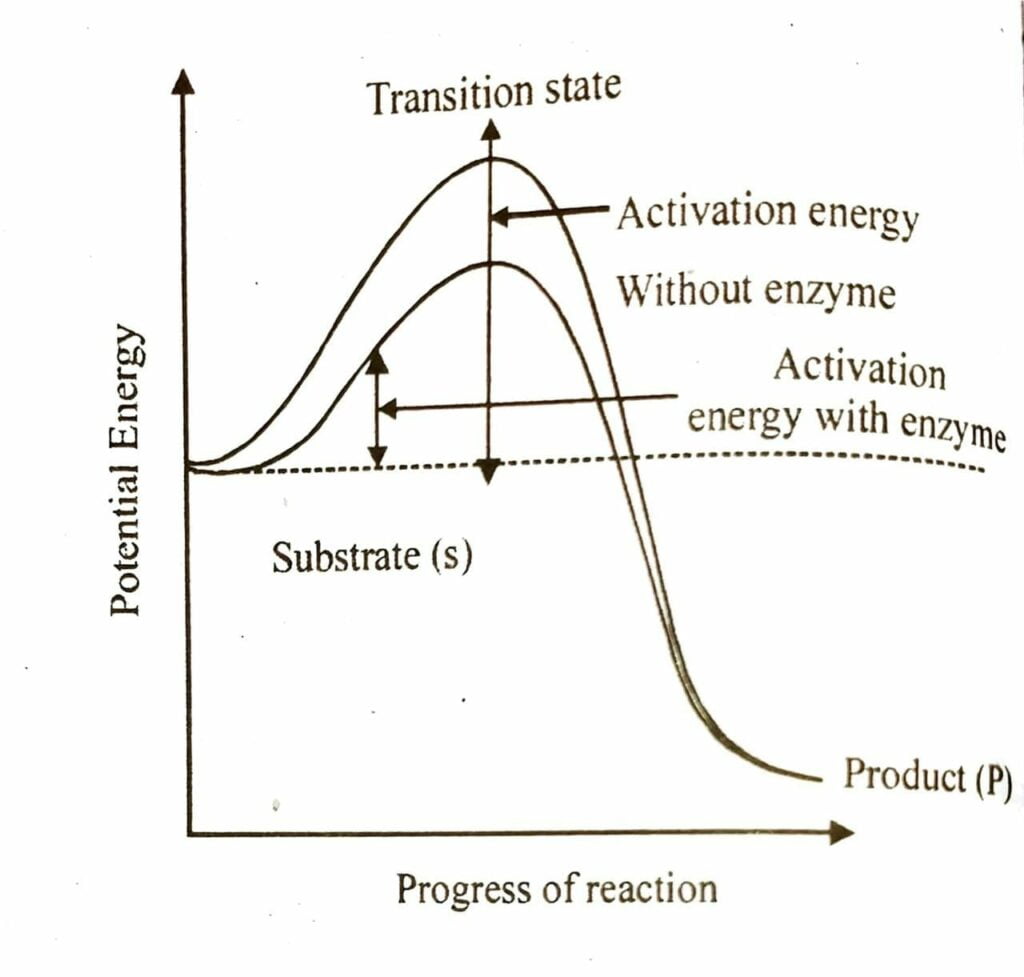
Q.15. Show the mechanism of enzyme action.
Ans :- Every enzyme (E) has a substrate (S) binding site in its molecule so that enzyme substrate complex is produced (ES). This complex is less stable and dissociates into its products (P) and the unchanged enzyme with an intermediate formation of enzyme product complex (EP).
The formation of ES complex is essential for catalysis.
E+S ⇒ ES → EP → E+P.
Firstly the substrate binds to the active site of the enzyme. The active site of the enzyme breaks the chemical bond of the substrate and a new enzyme product complex is formed. The enzyme releases the product of the reaction and the free enzyme is ready to bind to another molecule of the substrate again.
LONG ANSWER TYPE QUESTIONS
Q.1. What are polysaccharides ? Mention the names of important polysaccharide with their functions ?
Ans :- Polysaccharides are very complex carbohydrates. They are considered as polymers of monosaccharides. On hydrolysis they yield large number of monosaccharides. They are threads containing different monosaccharides as building blocks.
The most important polysaccharides are polymers of glucose.
Cellulose :- Cellulose is known as structural polysaccharide consisting of only one type of monosaccharide i.s glucose. It is a homopolymer. Paper made from pulp is cellulose.
Starch :- Starch is a complex carbohydrate. It occurs in many plants as storage food. The main sources of starch are rice, wheat, potato, corn etc. Man’s greatest source of carbohydrate is plant starch. It consists of large number of glucose molecules.
Glycogen :- It is commonly known as animal starch like starch, it is also a polymer of glucose units. It serves as a storage food in animals body.
Chitin :- It is a nitrogen containing polysaccharide. It occurs in the cell wall of fungi and exoskeleton of many arthropods.
Q.2. Explain the chemical structure of nucleic acid ?
Ans :- The structural unit forming a nucleic acid molecule are called nucleotide. A single nucleoside molecule is formed by the combination of 3 simple compounds a nitrogenous base, a pentose sugar and a phosphoric acid. On the basis of structure nitrogenous bases are classified into 2 types purine and pyrimidine. Adenine and Guanine are the prurine. Cytosine, Thymine and Uracil are the pyrimidine.
Pentose sugar are the monosaccharide containing five carbon atoms in their molecules. They are of 2 types ribose sugar and deoxyribose sugar. Ribose sugar is found in RNA and deoxyribose in DNA.
Nucleoside is the product of condensation of a nitrogenous base and a pentose sugar.
A nucleotide is the product of condensation of a nucleoside and phosphoric acid molecule.
Nucleotide are strong together to form a long chain called polynucleotide chain. The chemical bond formed between two nucleotide is known as phosphodiesterase bond. The adjacent nucleotide are connected together forming the sugar phosphate chain in which sugar and phosphate molecules are arranged in alternate fashion. The phosphate molecule of a nucleotide is joined to the third carbon atom (C³) of the deoxyribose of the next nucleotide. The nitrogenous base is attached to the first carbon atom (C¹) of the deoxyribose sugar.
DNA molecule consists of two polynucleotide chain, twisted about one another in the form of a coiled stair case like structure called helix. The two polynucleotides chains are joined together by their bases through weak hydrogen bonds. Thus the bases of one chain pair with those of the other chain.
Q.3. Who proposed the model of DNA ? What are the salient features of this model ?
Ans :- The model of DNA was given by watson and Crick in 1953, for which they were given Nobel prize in 1953, for which they were given Nobel prize in 1962. The salient features of this model are.
i) The DNA helix is formed by two polynucleotide strand. They are joined by third based through weak hydrogen bonds. The two strands are antiparallel i.e one proceeds in 3-5 direction and the others proceeds in 5-3 direction.
ii) The helix has the diameter of 20A° throughout its length.
iii) The helix makes one complete turn at every 34A°, along its length and hence since the internucleotide distance is 3.4A°, it consists of a stock of then nucleotide per turn.
(iv) The adjacent deoxyribonucleotide are joined in a chain by phosphodiester bond which link the 5 carbon atom of deoxyribose sugar of one nucleotide with the 3 carbon atom of the deoxyribose sugar of the next nucleotide unit.
v) The two strands are held together by weak hydrogen bond formed between pairs of purine and pyrimidine bases. They are formed in such a way that adenine base can bond only to thymine base by two hydrogen bonds and guanine can bond only to cytosine by three hydrogen bonds.
vi) Each strand appears like a helical staircase. Each step for ascent is represented by a pair of bases.
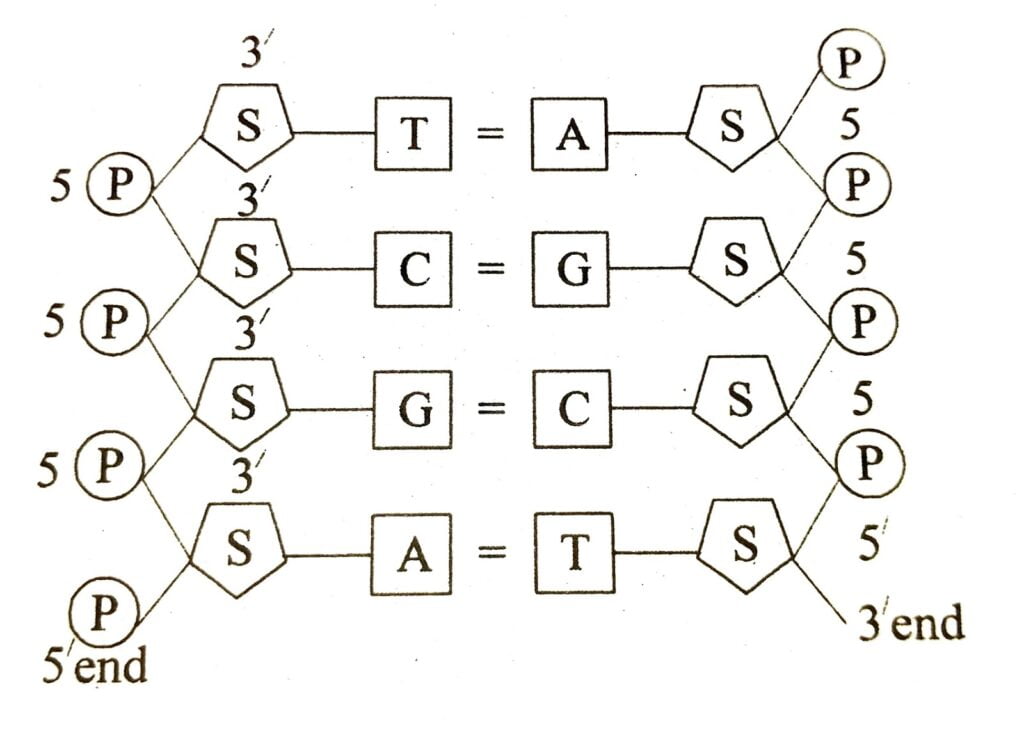
A part of DNA showing base pairing.
Q.4. Classify enzymes and describe any five of them.
Ans :- Enzymes are classified into different groups based on the type of reaction they catalyse.
Enzymes are divided into 6 classes :-
(i) Oxide-reductase/dehydrogenase :- Enzymes which catalyse oxido-reduction between two substrate S an S e.g.
S reduced +S oxidised S oxidised +S reduced.
ii) Transferase :- Enzyme catalysing a transfer of a group, G between a pair of substrate S and S e.g.
S-G+S S+S-G
iii) Hydrases :- Enzymes catalysing hydrolysis of ester, ether, peptide,
C-C, C-halede or P-n bonds.
iv) Lyases :- Enzymes that catalyse removal of groups from substrate by mechanism other than hydrolysis leaving double bonds.
X Y
1 1 → x -y + c = c
C – C
v) Ligase :- Enzyme catalysing the linking together of two compounds e.g enzymes which catalyse joining of
C-O, C-S, C-N, P-O etc bonds.
Q.5. Write a short note on co-factors.
Ans :- Some enzymes cannot act on their substrate unless there are certain assisting on-protein components called co factors. These are activators of enzyme e.g. Mn, Ni, Mg etc.
There are a number of cases in which the non protein constituent called co factors are bound to the enzyme to make the enzyme catalytically active. In these instances, the protein portion of the enzyme is called the apoenzyme. Three kinds of cofactor May be identified-prosthetic group, coenzyme and metal ions.
Prosthetic groups are organic compound and are distinguished from other cofactor in that they are tightly bound to the apoenzyme.
Coenzymes are also organic compounds but their association with the apoenzyme is also transient, usually occurring during the course of catalysis. Coenzymes serve as co factors in a number of different enzyme catalysed reaction. e.g. NAD.
A number of enzymes require metal ions for their activity which form coordination bonds with side chains at the active site and at the same time form one or more coordination bond with the substrate e.g zinc is a cofactor for the peridotitic enzyme carboxypeptidase.
Then the cofactor is removed from enzyme the catalytic activity is lost. This testifies that cofactor play a crucial role in the catalytic activity of the enzyme.

Hi, I’m Dev Kirtonia, Founder & CEO of Dev Library. A website that provides all SCERT, NCERT 3 to 12, and BA, B.com, B.Sc, and Computer Science with Post Graduate Notes & Suggestions, Novel, eBooks, Biography, Quotes, Study Materials, and more.


