Class 10 Science Chapter 1 Chemical Reaction and Equations The answer to each chapter is provided in the list so that you can easily browse throughout different chapters NCERT Class 10 Science Chapter 1 Chemical Reaction and Equations and select need one.
Class 10 Science Chapter 1 Chemical Reaction and Equations
Also, you can read the SCERT book online in these sections Solutions by Expert Teachers as per SCERT (CBSE) Book guidelines. These solutions are part of SCERT All Subject Solutions. Here we have given Assam Board Class 10 Science Chapter 1 Chemical Reaction and Equations Solutions for All Subjects, You can practice these here.
Chemical Reaction and Equations
Chapter – 1
GENERAL SCIENCE
Textual Questions and Answers:
Q.1. Why should a magnesium ribbon be cleaned before burning in air?
Ans: Because it quickly forms the white layer of magnesium oxide, which does not burn.
Q.2. Write the balanced equation for the following chemical reactions.
(i) Hydrogen + chloride → Hydrogen chloride
(ii) Barium chloride + Aluminium sulphate → Barium sulphate + Aluminium chloride
(iii) Sodium + water → Sodium hydroxide + Hydrogen.
Ans: (i) H₂+ Cl₂ → 2HCl
(ii) 3BaCl₂+ AI₂(SO₄)₃ → 3BaSO₄ + 2AICl₃
(iii) 2Na + 2H₂O → 2NaOH + H₂
Q.3. Write a balanced chemical equation with state symbols for the following reactions.
(i) Solutions of barium chloride and sodium sulphate in water react to give insoluble barium sulphate and the solution of sodium chloride.
(ii) Sodium hydrochloric acid solution (in water) to produce sodium chloride solution an water.
Ans: (i) Bacl₂ (aq) + Na₂SO₄ (aq) → BaSO₄ (s) + 2Nacl (aq)
(ii) NaOH (aq) + Hcl(aq) → Nacl(aq) + H₂O
Page No – 10
Q.1. A solution of substance ‘x’ is used for white-washing.
(i) Name the substance ‘x’ and write its formula.
(ii) Write the reaction of the substance ‘x’ named in (i) above with water.
Ans: (i) x’ is quick lime; CaO
(ii) CaO + H₂O → Ca(OH)₂
Q.2. Why is the amount of gas collected in one of the test tubes in Activity 1.7 double of the amount collected in the other? Name this gas.
Ans: Activity 1.7 depicts the process of electrolysis of water.
The decomposition of H2O compounds into H2 and O2 with the help of electricity is known as electrolysis of water.
The chemical equation for the electrolysis of water can be written as:
2H₂O → 2H₂ + O₂
The equation implies that water consists of two parts of hydrogen and one part of oxygen. It is evident that the amount of hydrogen gas collected in one test tube is double that of the oxygen collected in the other test tube. Therefore, the required name of the gas is hydrogen.
Page No -13
Q.1. Why does the colour of copper sulphate solution change when an iron nail is is kept immersed in it?
Ans: When an iron nail is placed in a copper sulphate solution, iron displaces copper from copper sulphate solution forming iron sulphate, which is green in colour.
Therefore, the blue colour of copper sulphate solution fades and green colour appears.
CuSO4 + Fe ⟶ FeSO4 + Cu
Q.2. Give an example of a double displacement reaction other than the one given in Activity 1.10.
Ans: When sodium carbonate reacts with calcium chloride, it forms a precipitate of calcium carbonate and sodium chloride. In this reaction, exchange of carbonate and chloride ions form two new compounds.
Na2CO3(aq) + CaCl2(aq) → CaCO3(aq) + 2NaCl(aq)
Sodium Carbonate Calcium carbonate Calcium carbonate Sodium chloride
This is double displacement reaction.
Q.3. Identify the substances that are oxidised and the substances that are reduced in the following reactions:
(i) 4Na(s) + O₂ (g) → 2Na₂O (s)
(ii) CuO (s) + H₂ (g) → Cu (s) + H₂O (l)
Ans: (i) Oxidised: Na(s), H₂(g)
(ii) Reduced: O₂(g), CuO(s)
Exercises Question Answer:
Q.1. Which of the statements about the reaction below are incorrect?
2PbO(s)+C(s) → 2Pb(s) + (Co₂(g)
(a) Lead is getting reduced.
(b) Carbon dioxide is getting oxidised.
(c) Carbon is getting oxidised.
(d) Lead oxide is getting reduced.
(i) (a) and (b)
(ii) (a) and (c)
(iii) (a), (b) and (c)
(iv) all
Ans: (i) (a) and (b)
Q.2. Fe₂O₃+ 2Al → Al₂O₃+ 2Fe The above reaction is an example of a
(a) combination reaction.
(b) double displacement reaction .
(c) decomposition reaction.
(d) displacement reaction.
Ans: (d) déplacement réaction.
Q.3. What happens when dilute hydrochloric acid is added to iron filling? Tick the correct answer.
(a) Hydrogen gas and iron chloride are produced.
(b) Chlorine gas and iron hydroxide are produced.
(c) No reaction takes place.
(d) Iron salt and water are produced.
Ans: (a) Hydrogen gas and iron chloride are produced.
Q.4. What is a balanced chemical equation? Why should chemical equations be balanced?
Ans: A chemical equations is balanced so that the numbers of atoms of each type involved in a chemical reaction are the same on the reactant and product sides of the equation.
The chemical equations must be balanced because the number of atoms of each element remains the same, before and after a chemical reaction.
Q.5. Translate the following statements into chemical equations and then balance them:
(a) Hydrogen gas combines with nitrogen to form ammonia-
(b) Hydrogen sulphide gas burns in air to give water and sulphur dioxide.
(c) Barium chloride reacts with aluminium sulphate to give aluminium chloride and a precipitate of barium sulphate.
(d) Potassium metal reacts with water to give potassium hydroxide and hydrogen gas.
Ans: (a) 3H₂(g) + N₂(g) → 2NH₃(g)
(b) 2H₂s(g) + 3O₂ → 2SO₂(g) + 2H₂O(e)
(c) 3BaCl₂(aq) + Al₂(SO₄)₃(aq) → 2AlCI₃(aq) + 3BaSO₄(s)
(d) 2K(s) + 2H₂O(l) → 2KOH (aq) + H₂(g)
Q.6. Balance the following chemical equations –
(a) HNO₃ + Ca(OH)₂ → Ca(NO₃)₂ + H₂O
(b) NaOH + H₂SO₄ → Na₂SO₄ + H₂O
(c) Nacl + AgNO₃ → Agcl + NaNO₃
(d) Bacl₂ + H₂SO₄ → BaSo₄ + HCI
Ans: (a) 2HNO₃ + Ca(OH)₂ → Ca(NO₃)₂ + 2H₂O
(b) 2NaOH + H₂SO₄ → Na₂SO₄ + 2H₂O
(c) Nacl + AgNO₃ → Agcl + NaNO₃
(d) Bacl₂ + H₂SO₄ → BaSO₄ + 2Hcl
Q.7. Write the balanced chemical equations for the following reactions:
(a) Calcium hydroxide + Carbon dioxide → Calcium Carbonate + water
(b) Zinc + Silver nitrate → Zinc nitrate + Silver
(c) Aluminium + Copper chloride → Aluminium Chloride + copper
(d) Barium chloride + Potassium sulphate → Barium sulphate + Potassium chloride.
Ans :- (a) Ca(OH)₂ + CO₂ → CaCO₃ + H₂O
(b) Zn + 2AgNO₃ → Zn(NO₃)₂ + 2Ag
(c) 2Al + 3CuCl₂ → 2AICI₃ + 3Cu
(d) BaCl₂ + K₂SO₄ → BaSo₄ + 2Kcl
Q.8. Write the balanced chemical equation for the following mule and identify the type of reaction in each case.
(a) Potassium bromide (aq) + Barium iodide (ag) → Potassium iodide (aq) + Barium bromide(s)
(b) Zinc carbonate (s) → Zinc oxide(s) + carbon di-oxide(g)
(c) Hydrogen(g) + chloringe(g) → Hydrogen chloride(g)
(d) Magnesium(s) + Hydrochloric acid(aq) → Magnesium chloride(aq) + Hydrogen (g)
Ans: (a) 2KBr (aq) + Bal₂ (aq) → KI(aq) + BaBr₂ (s)
Type: Double displacement reaction.
(b) ZnCO₃(s) → ZnO(s) + CO₂(g)
Type: Decomposition reaction.
(c) H₂(g) + Cl₂ (g) → 2HcI(g)
Type: Combination reaction.
(d) Mg(s) + 2Hcl(aq) → Mgcl₂ (aq) + H₂(g)
Type: Displacement reaction.
Q.9. What does one mean by exothermic and endothermic reactions? Give examples.
Ans: The chemical reactions that release heat energy are called exothermic reactions.
Example: C(g) + O2(g) → CO2(g) + Heat Energy
The chemical reactions in which heat energy is absorbed are called endothermic reactions.
Example: CaCO3 Heat → CaO + CO2
Q.10. Why is respiration considered an exothermic reactions? Explain.
Ans: During digestion, food is broken down into simpler substances. For example- rice, potatoes and bread contain carbohydrates. These carbohydrates are broken down to form glucose. This glucose combines with oxygen in the cells of our body and provides energy. This reaction is known as respiration. The chemical equation is-
C₆H₁₂ O₆ (aq) + 6O₂ (aq) → 6CO₂ (aq) + 6H₂O(e) + energy Thus respiration is an exothermic reaction.
Q.11. Why are decomposition reactions called the opposite of combination reactions? Write equations for these reactions.
Ans: Decomposition reactions are opposite of combination reactions. In a decomposition reaction, a single substance decomposes to give two or more substances.
For example:
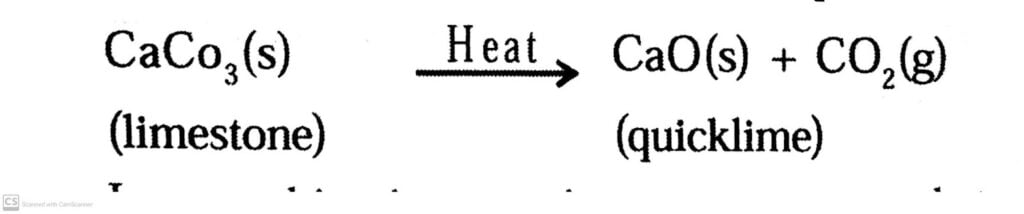
In a combination reaction two or more substances combine to form a new single substance.
For example: C(s) + O₂(g) → CO₂(g)
Q.12. Write one equation each for decomposition reactions where energy is supplied in the form of heat, light or electricity.
Ans:

Q.13. What is the difference between displacement and double displacement reactions? Write equations for these reactions.
Ans: Displacement reaction: A displacement reaction is a type of chemical reaction in which a more reactive element displaces a less reactive element from its compound or salt solution.
Example: When Zinc(Zn) metal is dipped in the Copper sulphate(CuSO4) solution, Zinc displaces the copper metal from its salt solution and forms Zinc sulphate (ZnSO4) and Copper is deposited at the bottom. CuSO4 (aq) + Zn(s) → ZnSO4(aq) + Cu(s)
Double displacement reaction: A double displacement reaction is a type of chemical reaction in which two compounds react by exchanging their ions to form new compounds. It is typically identified by the formation of a product that is a precipitate.
Example: When Sodium sulphate (Na2SO4) is treated with Barium chloride (BaCl2), it will lead to the formation of Barium sulphate(BaSO4) and Sodium chloride (NaCl).
Na2SO4(aq)+BaCl2(aq) → BaSO4(s)+2NaCl(aq)
Q.14. In the refining of silver, recovery of silver from silver nitrate solution involved displacement by copper metal. Write down the reaction involved.
Ans: AgNO₃ (aq) + Cu(s) → CuNO₃ (aq) + Ag (s)
Q.15. What do you mean by a precipitation reaction? Explain by giving examples.
Ans: A precipitate is an insoluble substance.
A reaction in which any insoluble solid precipitate is formed is called Precipitation Reaction.
For example:
- When Sodium Sulphate solution is mixed with Barium Chloride solution It forms Barium Sulphate and Sodium Chloride solution.
In this reaction, a white precipitate of Barium Sulphate is formed.
For example: Na₂SO₄ (aq) + Bacl₂ (aq) → BaSO₄ (s) + 2Nacl(aq)
- When Potassium Iodide solution is added to Lead Nitrate solution A yellow precipitate of Lead Iodide is formed along with Potassium Nitrate solution
KI + Pb(NO3 ) 2 → KNO3 + PbI2
Q.16. Explain the following in terms of gain or logs of oxygen with two example reach –
(a) Oxidation.
(b) Reduction.
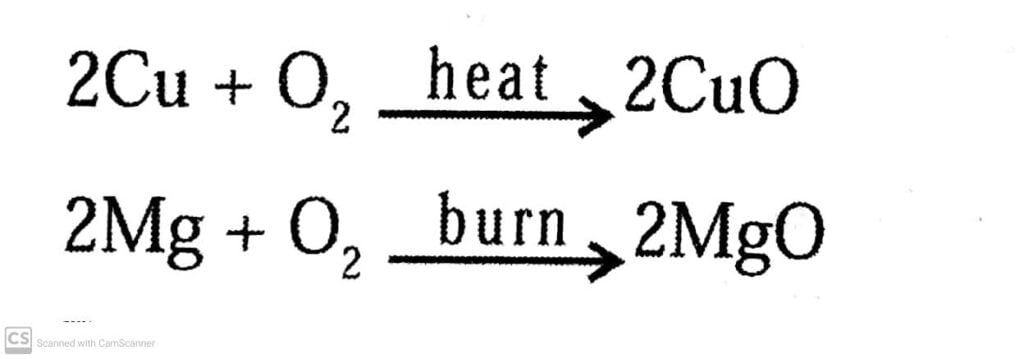
Ans: (a) Oxidation: Oxidation is a process in which a chemical substance changes because of the addition of oxygen.
Example:
(b) Reduction: The chemical reactions in which a substance losses oxygen is called reduction.
Example: ZnO + C → Zn + CO
Cuo + H₂ → Cu + H₂O
Q.17. A shiny brown colured element ‘x’ on heating in air becomes black in colour. Name the element ‘x’ and black coloured compound formed.
Ans: The shiny elements are metals. The brown coloured metal is copper. Hence ′X′ is copper and the black coloured compound formed is Copper oxide.
The reaction involved is:
2Cu(s)+O2(g)→CuO(s)
Q.18. Why do we apply paint on iron articles?
Ans: Iron articles are coated with paint to prevent air and moisture from coming in contact with iron and hence, to protect it from rusting.
Q.19. Oil and fat containing food items are flushed with nitrogen. Why?
Ans: The main purpose of flushing Nitrogen into food packets that contain oil and fat items is to prevent Rancidity which occurs when the oil or fat reacts with the oxygen letting out an unpleasant smell and taste. Therefore, by flushing Nitrogen, an unreactive surrounding is created thus preventing rancidity.
Q.20. Explain the following terms with an example each:
(a) corrosion.
(b) Rancidity.
Ans: (a) Corrosion: Corrosion definition states that it is a natural process that causes the transformation of pure metals into undesirable substances when they react with substances such as water or air. This reaction causes damage or disintegration of metal as it interfaces with the environment and gradually spreads to the entire bulk of the metal.
(b) Rancidity: In chemistry, a state known as rancidity, also known as rancidification, is brought on by the aerial oxidation of unsaturated fat found in foods and other items that have off-putting smells or aromas.
Unsaturated parts of a fatty substance can become hydroperoxides when exposed to sunlight, which then decompose into esters, volatile aldehydes, ketones, alcohols, and hydrocarbons, some of which have foul smells. Using some preventative measures, rancidity can also be avoided.
Multiple Choice Questions:
Q.1. Identify the double decomposition reactions.
(a) Hydrogen burns in air.
(b) Electrolysis of water.
(c) Digestion of food.
(d) Addition of dilute sulphuric acid to BaCl₂ solution.
Ans: (d) Addition of dilute sulphuric acid to BaCl₂ solution.
Q.2. Choose the combination reaction.
(a) Electric current is passed through water.
(b) Hydrogen burns in oxygen.
(c) Hydrogen peroxide is exposed to sunlight.
(d) Digestion of food in our body.
Ans: (b) Hydrogen burns in oxygen.
Q.3. Food items on exposures to atmosphere become rancid due to the process of.
(a) Oxygen.
(b) Reduction.
(c) Corrosion.
(d) Hydrogenation.
Ans: (a) Oxidation.
Q.4. What is not true in a balanced chemical equation?
(a) Number of atoms of different elements on both sides are equal.
(b) Mass of both sides are equal.
(c) Number of charged ions on both sides are equal.
(d) Total of charges on both sides are equal.
Ans: (c) Number of charged ions on both sides are equal.
Q.5. Respiration is a chemical change because.
(a) Energy is evolved.
(b) There is decrease in temperature.
(c) There is increase in temperature.
(d) There is change in state.
Ans: (a) Energy is evolved.
Q.6. In a chemical equation, what is avoided?
(a) Symbols.
(b) Formula.
(c) Number of atoms. And
(d) Words.
Ans: (d) Words.
Q.7. Choose the double displacement reaction
(a) X + YZ → XY + Z
(b) X + Y → XY
(c) XYZ → YZX
(d) XY + AB → XA + YB
Ans: (d) XY + AB → XA + YB
Q.8. Which is true for an oxidation reaction?
(a) Addition of oxygen.
(b) Removal of oxygen.
(c) Removal of hydrogen.
(d) Addition of hydrogen.
(i) (a) and (c)
(ii) (a) and (d)
(iii) (a) and (b)
(iv) (b) and (d)
Ans: (i) (a) and (c)
Q.9. Which one of the following can be a non-redox reaction?
(a) Combination.
(b) Decomposition.
(c) displacement.
(d) Double displacement.
Ans: (d) Double déplacement.
Q.10. Identify the double decomposition reactions.
(a) Hydrogen burns in air.
(b) Electrolysis of water.
(c) Digestion of food.
(d) Addition of dilute sulphuric acid to BaCl₂ solution.
Ans: (d) Addition of dilute sulphuric acid to BaCl₂ solution.
Q.11. Choose the combination reaction.
(a) Electric current is passed through water.
(b) Hydrogen burns in oxygen.
(c) Hydrogen peroxide is exposed to sunlight.
(d) Digestion of food in our body.
Ans: (b) Hydrogen burns in oxygen.
Q.12. Food items on exposures to atmosphere become rancid due to the process of.
(a) Oxygen.
(b) Reduction.
(c) Corrosion.
(d) Hydrogenation.
Ans: (a) Oxidation.
Q.13. What is not true in a balanced chemical equation?
(a) Number of atoms of different elements on both sides are equal.
(b) Mass of both sides are equal.
(c) Number of charged ions on both sides are equal.
(d) Total of charges on both sides are equal.
Ans: (c) Number of charged ions on both sides are equal.
Q.14. Respiration is a chemical change because.
(a) Energy is evolved.
(b) There is decrease in temperature.
(c) There is increase in temperature.
(d) There is change in state.
Ans: (a) Energy is evolved.
Q.15. In which of the following experiments, residue left behind is reddish brown?
(a) Heating of coper powder in air.
(b) Heating of ferrous sulphate crystals.
(c) Heating of magnesium in air.
(d) Heating of solid calcium carbonate.
Ans: (b) Heating of ferrous sulphate crystals.

Hi, I’m Dev Kirtonia, Founder & CEO of Dev Library. A website that provides all SCERT, NCERT 3 to 12, and BA, B.com, B.Sc, and Computer Science with Post Graduate Notes & Suggestions, Novel, eBooks, Biography, Quotes, Study Materials, and more.



I want to buy SEBA class 10 science question answer solution pdf in English medium. Kindly help me!!!!!!
coming soon