NIOS Class 10 Science and Technology Chapter 27 Air and Water Solutions to each chapter is provided in the list so that you can easily browse through different chapters NIOS Class 10 Science and Technology Chapter 27 Air and Water and select need one. NIOS Class 10 Science and Technology Chapter 27 Air and Water Question Answers Download PDF. NIOS Study Material of Class 10 Science and Technology Notes Paper 212.
NIOS Class 10 Science and Technology Chapter 27 Air and Water
Also, you can read the NIOS book online in these sections Solutions by Expert Teachers as per National Institute of Open Schooling (NIOS) Book guidelines. These solutions are part of NIOS All Subject Solutions. Here we have given NIOS Class 10 Science and Technology Chapter 27 Air and Water, NIOS Secondary Course Science and Technology Solutions for All Chapters, You can practice these here.
Air and Water
Chapter: 27
INTEXT QUESTIONS 27.1
1. Which properties of gold allows it to make ornaments?
Ans: Malleability and ductility.
2. Name a few metals which are found in free state?
Ans: Gold, silver and platinum.
3. Metals are generally very hard. Name the metal which is soft like wax?
Ans: Sodium.
4. Name a non-metal which is a good conductor of electricity.
Ans: Graphite (an allotropic form of carbon).
5. Name two metals which show malleability and ductility.
Ans: Gold and aluminium.
INTEXT QUESTIONS 27.2
1. Under what conditions there are more chances for iron to be rusted?
Ans: Iron will be rusted if there is oxygen (air) and water.
2. Why metals are electropositive but non-metals are electronegative in nature?
Ans: Metals have a tendency to lose electrons and get converted into a positive ion and therefore are electropositive. Non-metals have a tendency to take electrons and get converted into a negatively charged ion and therefore are electronegative.
3. Name a metal oxide which reacts with an acid as well as with a bases?
Ans: Al2O3 and ZnO are amphoteric in nature and react with acid and base.
4. When zinc reacts with sodium hydroxide what is the product? Write the equation.
Ans: Sodium zincate is formed.
Zn(s) + 2NaOH(aq) ⎯⎯→ Na2ZnO2(aq)
5. Write the formula of rust.
Ans: Fe2O3.xH2O.
INTEXT QUESTIONS 27.3
1. What will happen if you keep a solution of copper (II) sulphate in an iron vessel? Explain the observation and give suitable explanation.
Ans: Iron will react with copper (II) sulphate and after sometimes a hole will be formed in the bottom of iron vessel. Reaction will be as following
Fe(s) + CuSO4(aq) ⎯⎯→ FeSO4(aq) + Cu(s).
2. What will happen if you keep a solution of silver nitrate in a copper vessel? Explain the observation.
Ans: When silver nitrate is kept in copper vessel, copper will replace silver as copper is above silver in the activity series and a hole is expected in the bottom of copper vessel.
Reaction will be as following:
Cu(s) + 2AgNO3(aq) ⎯⎯→ 2Ag(s) + Cu(NO3)2(aq).
3. An element reacts with oxygen to form an oxide which dissolves in water to form a solution that turns red litmus blue. The oxide dissolves in dil. HCI. Identify the element as metal or non-metal.
Ans: Since oxide of the element turn read litmus blue therefore that must ba a basic oxide. This is further supported by dissolution of oxide in HCl. Basic oxide will be formed by a metal. Therefore element must be a metal.
4. Give an example of a metal which.
(a) is a liquid at room temperature.
Ans: Hg (mercury) is metal which is liquid at room temperature.
(b) can be easily cut with a knife.
Ans: Sodium metal can be cut easily with knife.
(c) is the best conductor of electricity.
Ans: Silver is best conductor of electricity.
(d) poorest conductor of electricity.
Ans: Iron is the poorest conductor of electricity.
5. Write the formula of the oxide of magnesium formed on burning of magnesium ribbon in oxygen?
Ans: When magnesium burns it forms magnesium oxide
2Mg(s) + O2 (g) ⎯⎯→ 2MgO(s).
6. Name the hydroxide of magnesium formed when magnesium oxide reacts with hot water?
Ans: MgO(s) + H2O(l) (hot) ⎯⎯→ 2Mg(OH)2(aq).
7. What happens when sodium metal reacts with water in cold? Write the reaction for the same?
Ans: 2Na(s) + H2O(l) ⎯⎯→ 2NaOH(aq).
8. Define activity series of metals? Write a reaction when zinc granules are added to copper sulphate solution?
Ans: When the metals are arranged in the decreasing order of their reactivity, a series is obtained, this series is called activity series. Metals in the upper position of the series can replace the metal in lower position from their aqueous solution. When zinc granules are added to copper sulphate solution, reaction will be as following.
Zn(s) + CuSO4(aq) ⎯⎯→ ZnSO4(aq) + Cu(s).
Zn is above copper in activity series therefore it (Zn) will replace copper from the solution.
9. What is the difference between ‘mineral’ and ‘ore’?
Ans: Naturally occuring homogeneous inorganic substances are called minerals. But those minerals from which metals can be extracted profitability are called ores. every ore is a mineral but every mineral cannot be an ore.
| TERMINAL EXERCISES |
1. Name two precious metals used in making ornaments and write names of two important properties of these metals.
Ans: Two precious metals used in making ornaments and write names of two important properties of these metals are mentioned below:
(i) gold.
(ii) silver.
2. Name two non-metals which are commonly available and name their two important properties.
Ans: Two non-metals which are commonly available and name their two important properties are mentioned below:
(i) Non-metals cannot be hammered into thin sheets.
(ii) Non-metals are not ductile.
3. Write four physical properties of metals.
Ans: Four physical of metals are mentioned below:
(i) Metals are malleable and ductile.
(ii) Metals are good conductors of heat and electricity.
(iii) Metals are lustrous (shiny) and can be polished.
(iv) Metals are solids at room temperature (except mercury, which is liquid).
Metals are tough and strong.
4. Write four physical properties of nonmetals.
Ans: Four physical of non metals are mentioned below:
(i) Non-metals are brittle.
(ii) Non-metals are bad conductors of heat and electricity (except graphite).
(iii) Non-metals are non-lustrous (dull) and cannot be polished (except iodine).
(iv) Non-metals may be solids, liquids or gases at room temperature.
Non-metals are neither tough nor strong.
5. How would you differentiate between a metals and a non-metal.
Ans:
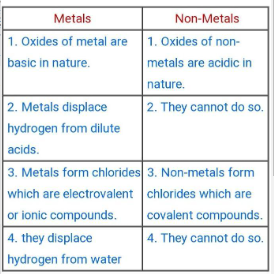
6. Write reaction of metals with the following:
(a) water.
Ans: (a) Na + H2O —> NaOH.
(b) air or oxygen.
Ans: 2Zn + O2 —> 2ZnO.
(c) Acids.
Ans: Zn + H2SO4 —> ZnSO4.
7. What are three types of oxides of metals?
Ans: The three types of oxides of metals are:
(i) monoxide.
(ii) Dioxide.
(iii) Trioxide.
8. Name four uses of metals.
Ans: Four uses of metals:
(i) Construction.
(ii) Electronic devices.
(iii) Transportation.
9. Write four uses of non-metals.
Ans: Four uses of non-metals are mentioned below:
(i) Some non – metals and their uses are listed below:
(ii) Oxygen is used for breathing.
(iii) Chlorine is used for purifying water.
(iv) Phosphorus is used in crackers.
10. Define the following:
(a) Brittleness.
Ans: Brittleness is a material’s tendency to easily break, crack, or snap. Brittleness can occur in metals, ceramics, plastics, glass, and composite materials
(b) Sonorous nature.
Ans: The sonorous nature of metals means that they are able to make a ringing sound.
11. Name two metals which are most malleable and ductile. Also define:
Ans: Two metals which are most malleable and ductile are:
Malleability describes a material’s ability to be hammered, flattened, or pressed into thin sheets without breaking. Imagine taking a piece of metal and pounding it with a hammer – a highly malleable metal would spread out and become thinner and wider easily.
(ii) Gold.
(ii) silver.
(a) Malleability.
Ans: Malleability describes a material’s ability to be hammered, flattened, or pressed into thin sheets without breaking. Imagine taking a piece of metal and pounding it with a hammer – a highly malleable metal would spread out and become thinner and wider easily.
(b) Ductility.
Ans: Ductility refers to a material’s capacity to be drawn into thin wires. Think of stretching a piece of metal into a long, thin strand. A highly ductile metal would allow this stretching without snapping.
(c) Brittleness.
Ans: Brittleness is the opposite of malleability and ductility. Brittle materials break or shatter easily under stress, with little to no bending or deformation.
For example: if you try to bend a piece of ceramic tile, it will likely snap instead of bending.
(d) Tensile Nature.
Ans: Tensile strength refers to a material’s ability to resist pulling forces that tend to stretch it. It’s a measure of how much stress a material can withstand before breaking. So, while ductility tells us how easily a material can be pulled into a wire, tensile strength tells us how strong that wire will be.
12. Write uses of the following metals:
(a) Pt.
Ans: Pt is used in making ornaments and in catalysts.
(b) Au.
Ans: Au is used in making ornaments.
(c) Na.
Ans: Na for the making of table salt.
(d) Ag.
Ans: Ag is used as conductors in electrical transmission.
(e) Ni.
Ans: Ni is used as a catalyst.
13. What is corrosion? How will you prevent it?
Ans: Corrosion is the gradual deterioration of metals caused by the action of air, moisture, or a chemical reaction (such as an acid) on their surface. Rusting of iron, or the forming of a brown flaky material on iron objects when exposed to moist air, is the most common example of metal corrosion.
For the preventing or the corrosion we must amalgam it
14. You are provided atomic number of metal X, Y and Z (not real names) in the following table. Based on electronic configuration predict whether they fall in the category of metal or non-metal.
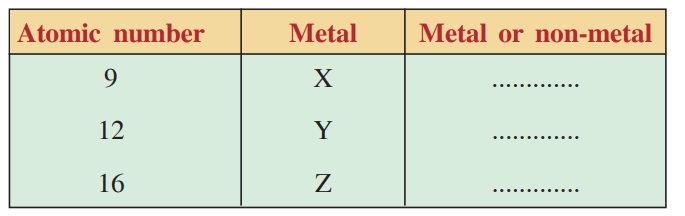
Ans:
| Atomic number | Metal | Metal or non-metal |
| 9 | x | Non metal |
| 12 | y | Metal |
| 16 | z | Non metal |
15. Complete and balance the following reactions:
(i) Mg(s) + H2SO4(aq) ⎯⎯→ …………… + ……………
Ans: MgSO4 and H2.
(ii) Fe(s) + H2O(steam) ⎯⎯→ …………… + ……………
Ans: Fe2O3 and 3H2.
(iii) Na(s) + H2O(l) ⎯⎯→ …………… + ……………
Ans: 2NaOH and H2.
16. Write names and formulas of different oxides of nitrogen.
Ans: The different formula of oxides of nitrogen is NO2, N2O, N2O5.
17. Which one of the following oxides is not acidic?
(a) CO.
(b) CO2.
(c) SO2.
(d) SO3.
Ans: (a) CO.
18. Write at least one important use of the following chemicals
(a) H3PO4.
(b) H2SO4.
(c) NH3.
(d) Water gas.
Ans: (a) H3PO4.
19. Identify the non-metal from the following which is used for fungus control in agriculture:
(a) Phosphorus.
(b) Sulphur.
(c) Iodine.
Ans: (a) Phosphorus.
20. Which of the following metals is not used in making cell/battery?
(a) Zn.
(b) Pb.
(c) Hg.
(d) Na.
Ans: (d) Na.
21. Which of the following non-metals is a liquid at room temperature
(a) Bromine.
(b) Phosphorous.
(c) Sulphur.
(d) Iodine.
Ans: (a) Bromine.
22. Complete the following reactions (i) Al2O3(s) + ⎯⎯→ 2NaAlO2 + H2O(l) (Sodium Aluminate)
Ans: 2NaOH.
(ii) CaO(s) + …………. ⎯⎯→ Ca(OH)2.
Ans: H2O.
(iii) Sn(s) + ………….+ H2O(l) ⎯⎯→ Na2SnO3
(Sodium stannate)
Ans: H2O.

Hi! my Name is Parimal Roy. I have completed my Bachelor’s degree in Philosophy (B.A.) from Silapathar General College. Currently, I am working as an HR Manager at Dev Library. It is a website that provides study materials for students from Class 3 to 12, including SCERT and NCERT notes. It also offers resources for BA, B.Com, B.Sc, and Computer Science, along with postgraduate notes. Besides study materials, the website has novels, eBooks, health and finance articles, biographies, quotes, and more.



