Class 10 Science Chapter 4 Carbon and its compounds The answer to each chapter is provided in the list so that you can easily browse throughout different chapters NCERT Class 10 Science Chapter 4 Carbon and its compounds and select need one.
Class 10 Science Chapter 4 Carbon and its compounds
Also, you can read the SCERT book online in these sections Solutions by Expert Teachers as per SCERT (CBSE) Book guidelines. These solutions are part of SCERT All Subject Solutions. Here we have given Assam Board Class 10 Science Chapter 4 Carbon and its compounds Solutions for All Subjects, You can practice these here.
Carbon and its compounds
Chapter – 4
GENERAL SCIENCE
Textual Questions and Answers:
Page – 61 Q.1. What would be the electron dot structure of carbon dioxide which has the formula CO₂?
Ans:

Carbon dioxide is a three-atom covalent molecule, with carbon surrounded by two oxygen atoms. Carbon and oxygen both have p orbitals that can interact due to their symmetry compatibility. According to the valence bond theory, carbon has four bonds and oxygen has two.
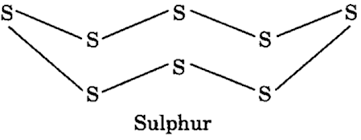
Q.2. What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur? [Hint: the eight atoms of sulphur are joined together in the form of a ring]
Ans: The atomic number of sulfur is 16 and its electronic configuration is 2,8,6.
The sulfur atom has six valence electrons. S8 is the chemical formula for a sulfur molecule, and each sulfur atom is bonded to identical atoms on each side by single covalent connections, completing its octet.
Page – 67
Q.1. Calculate the difference in the formulae and molecular masses for
(a) CH₃ OH and C₂H₅OH.
(b) C₂H₅OH and C₃H₇OH. and
(c) C₃H₇OH and C₄H₉OH.
Ans: (a) difference in formula is – CH₂ difference in molecular mass is 14 amu.
(b) difference in formula is – CH₂ difference in molecular mass is 14 amu.
(c) difference in formula is – CH₂ difference in molecular mass is 14 amu.
Q.2. Is there any similarity in these three?
Ans: Similarity observed in above three compounds are as follows:
(1) Difference in molecular formulae of these compounds is CH2.
(2) Difference in molecular masses of these compounds is 14u.
(3) In the nomenclature of these compounds, a common suffix -ol is written.
Q.3. Arrange these alcohols in the order of increasing carbon atoms to get a family. Can we call this family a homologous series?
Ans: CH₃OH, C₂H₅OH, C₃H₇OH, C₄H₉OH. This is a homologous series of alcohols.
Q.4. Generate the homologous series for compounds containing up to four carbons for the other functional groups given in table 4.3.
Ans: Homologous series of Halides – CH₃CI, C₂H₅CI, C₃H₇CI, C₄H₉Cl
Homologous series of Aldehydes – CH₃CHO, C₂H₅CHO, C₃H₇CHO
Homologous series of carboxylic acids – CH₃COOH, C₂H₅COOH, C₃H₇COOH
Page 68-69
Q.1. How many structural isomers can you draw for pentane?
Ans: Three structural isomers of pentane are:
(i) n – pentane:

(ii) Iso – Pantane:
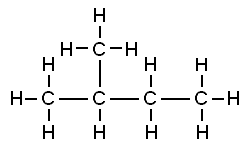
(iii) Neopentane:

Q.2. What are the two properties of carbon which lead to the huge number of carbon compounds we see around us?
Ans: (i) Catenation.
(ii) Tetravalency.
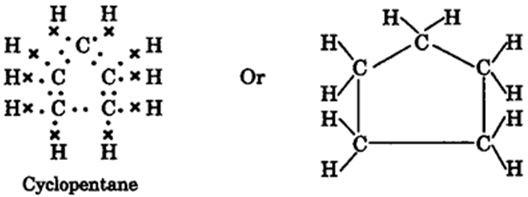
Q.3. What will be the formula and electron dot structure of cyclopentane?
Ans: The chemical formula for cyclopentane is C5H10, and it is an acyclic molecule.
Structure:
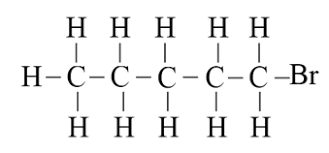
Q.4. Draw the structure for the following compounds:
(i) Ethanoic acid.
(ii) Bromopentane.
(iii) Butanone.
(iv) hexanol.
Ans: (i) Ethanoic acid:

(ii) Bromopentane:
(iii) Butanone:
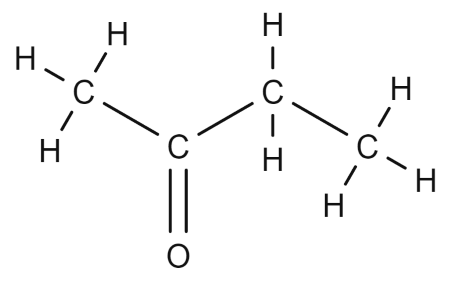
(iv) Hexanal:

There are three structural isomers for bromopentane depending on the position of Br at carbon 1, 2, 3.
There are:
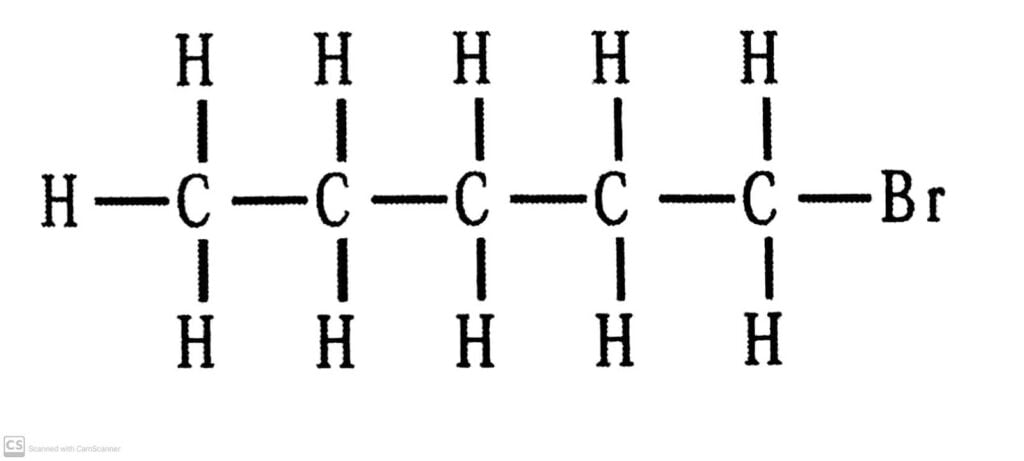
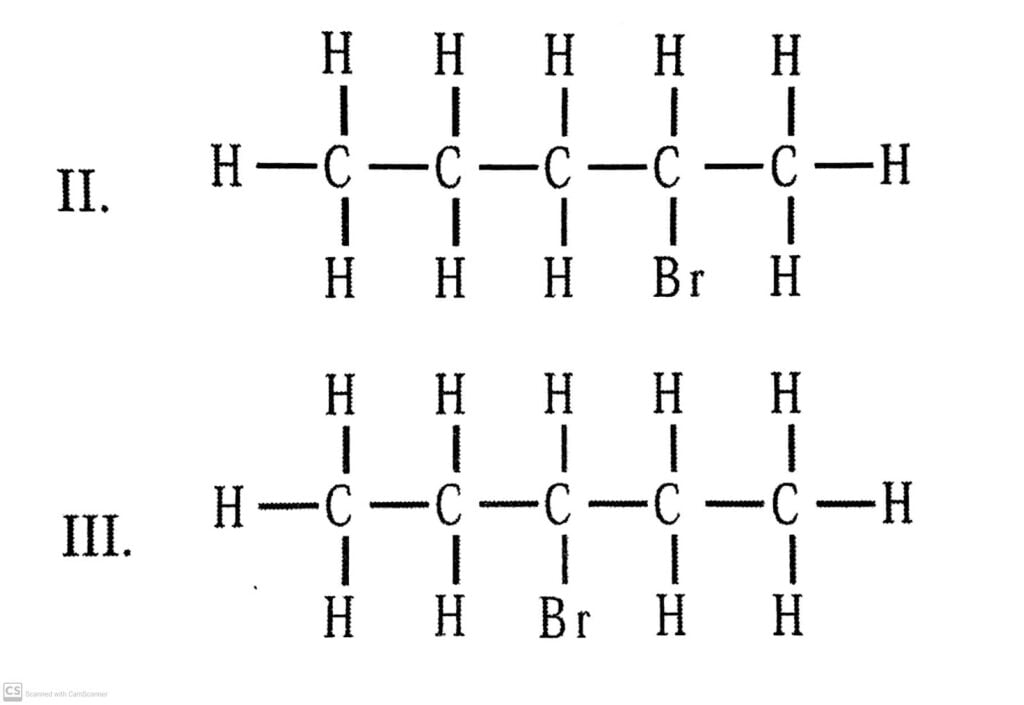
Q.5. How would you name the following compounds?
(i) CH₃-CH₂-Br
(ii)
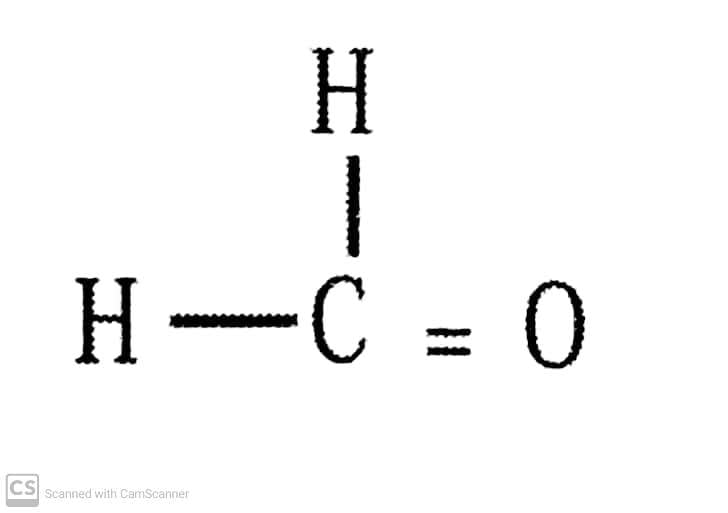
(iii)
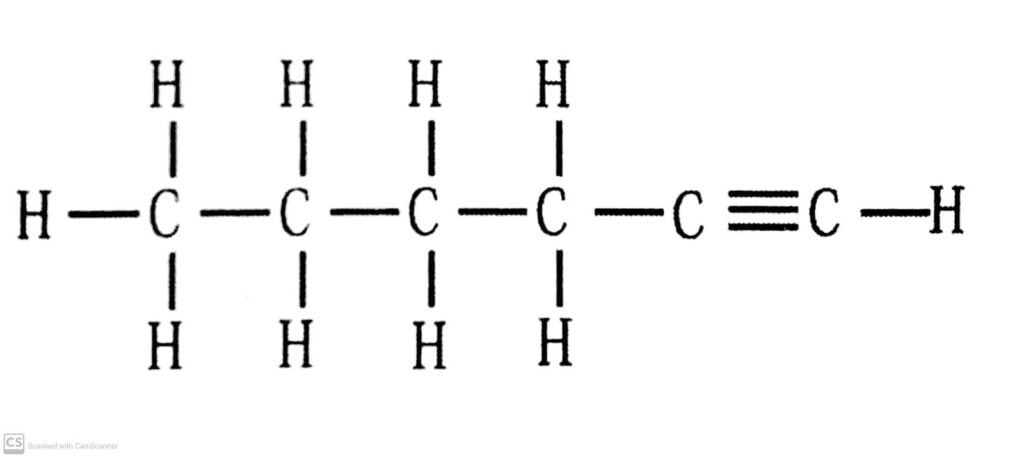
Ans: (i) Bromoethane.
(ii) Methanal.
(iii) 1, Hexyne.
Page – 71
Q.1. Why is the conversion of ethanol to ethanoic acid an oxidation reaction?
Ans: Alkaline potassium permanganate or acidified potassium dichromate are oxidising ethanol to ethanoic acid, that is adding oxygen. So conversion of ethanol to ethanoic acid an oxidation reaction.
Q.2. A mixture of oxygen and ethyne is burnt for welding. Can you tell why a mixture of ethyne and air is not used?
Ans: The oxygen-ethyne flame is extremely hot and produces a very high temperature which is used for welding metals. A mixture of ethyne and air is not used for welding because burning of ethyne in air produces a sooty flame due to incomplete combustion, which is not hot enough to melt metals for welding. But a mixture of ethyne and oxygen gives enough heat that can be used for welding.
Page – 74
Q.1. How would you distinguish experimentally between an alcohol and a carboxylic acid?
Ans: On reaction with Sodium Carbonate, Carboxylic acids produces carbon dioxide gas which turns lime water milky whereas alcohols do not give this reaction. This experiment can be used to distinguish an alcohol and carboxylic acid.
Reaction of Carboxylic acid with sodium carbonate:
2CH3COOH + Na2CO3 → 2CH3COONa + H2O + CO2
Q.2. What are oxidising agent?
Ans: Oxidising Agent.
Page – 76
Q.1. Would able to check if water is hard by using a no. detergent?
Ans: Detergents cannot be used to check the hardness of the water, as they lather easily in both soft and hard water. However, the hardness of water can be detected using soaps, as soaps don’t lather easily in hard water.
Q.2. People use a variety of methods to wash cloths, usually after adding the soap, they ‘beat’ the cloths on a stone, or beat it with a paddle, scrub with a brush or the mixture is agitated in a washing machine why is agitation necessary to get clean clothes?
Ans: It is necessary to agitate to get clean clothes because the soap micelles which entrap oily or greasy particles on the surface of dirty cloth have to be removed from its surface. When the cloth washed in soap solution is agitated or beaten, the micelles containing any or greasy dirt particles get removed from the surface of dirty cloth and go into water. And the dirty cloth gets cleaned.
EXERCISES
Q.1. Ethane, with the molecular formula C₂H₆ has
(a) covalent bonds.
(b) 7 covalent bonds.
(c) 8 covalent bonds.
(d) 9 covalent bonds.
Ans. (b) 7 covalent bonds.
Q.2. Butane is a four carbon compound with the fundamental group:
(a) carboxylic acid.
(b) aldehyde.
(c) ketone.
(d) alcohol.
Ans: (c) ketone.
Q.3. While cooking, if the bottom of the vessel is getting blackened on the outside, it means that:
(a) The food is not cooked completely.
(b) The fuel is not burning completely.
(c) The fuel is wet.
(d) The fuel is burned completely.
Ans: (b) The fuel is not burning completely.
Q.4. Explain the nature of the covalent bond using the bond formation in CH₃CI.
Ans: Covalent bond is formed by sharing of electrons so that the combining atom complete their outermost shell. In CH₃CI, this happens as follows.
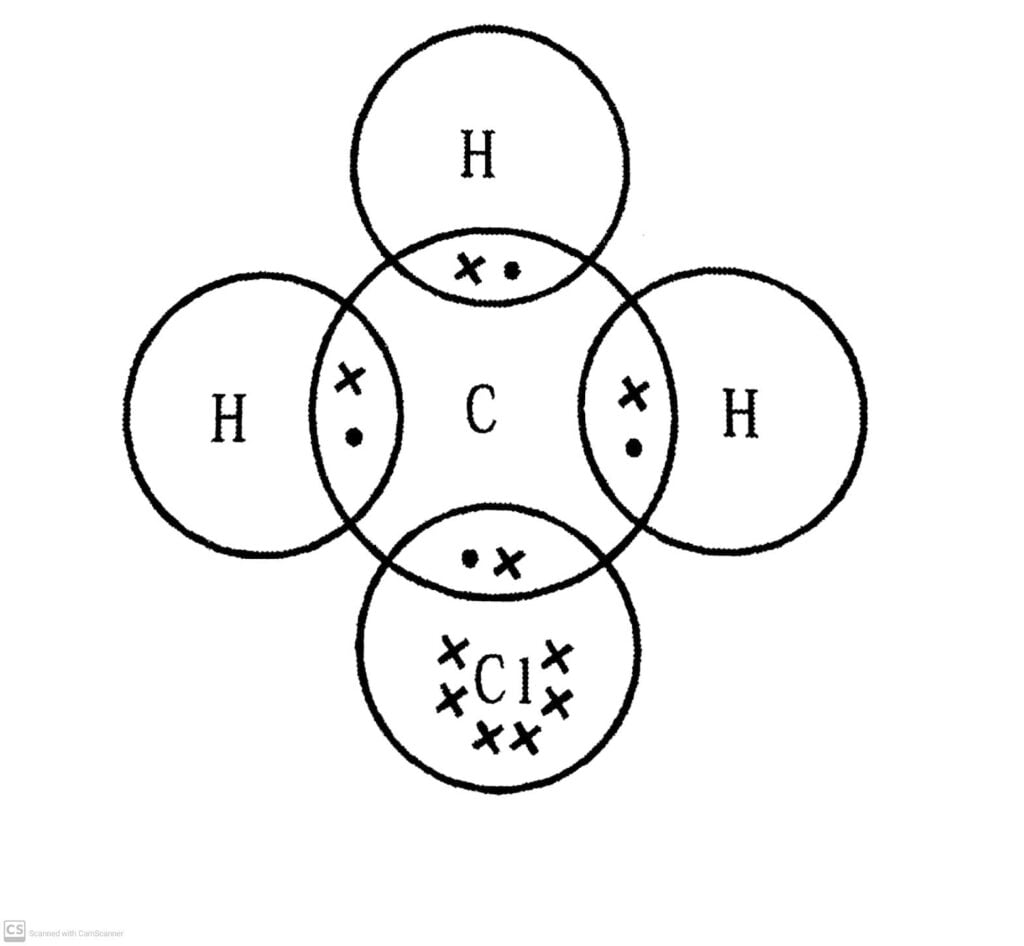
There hydrogen atoms complete their outermost shells by sharing three electrons of carbon atoms. Chlorine completes its outermost shell by sharing its one out of seven electrons with one electron of carbon atom. Thus carbon atom shares in all its four electrons with three of three hydrogen atoms and one of chlorine atoms and completes its outer shell.
Q.5. Draw the electron dot structures for
(a) ethanoic acid
(b) H₂S
(c) propanone
(d) F₂
Ans: (a) Ethanoic acid:

(b) H₂S
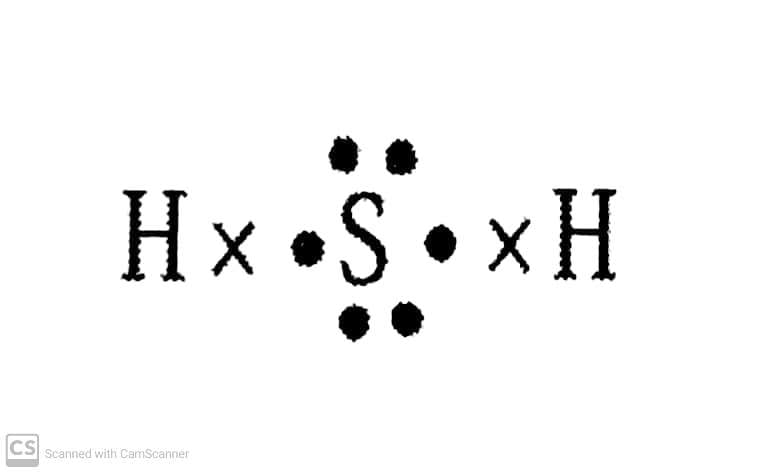
(c) Propanone
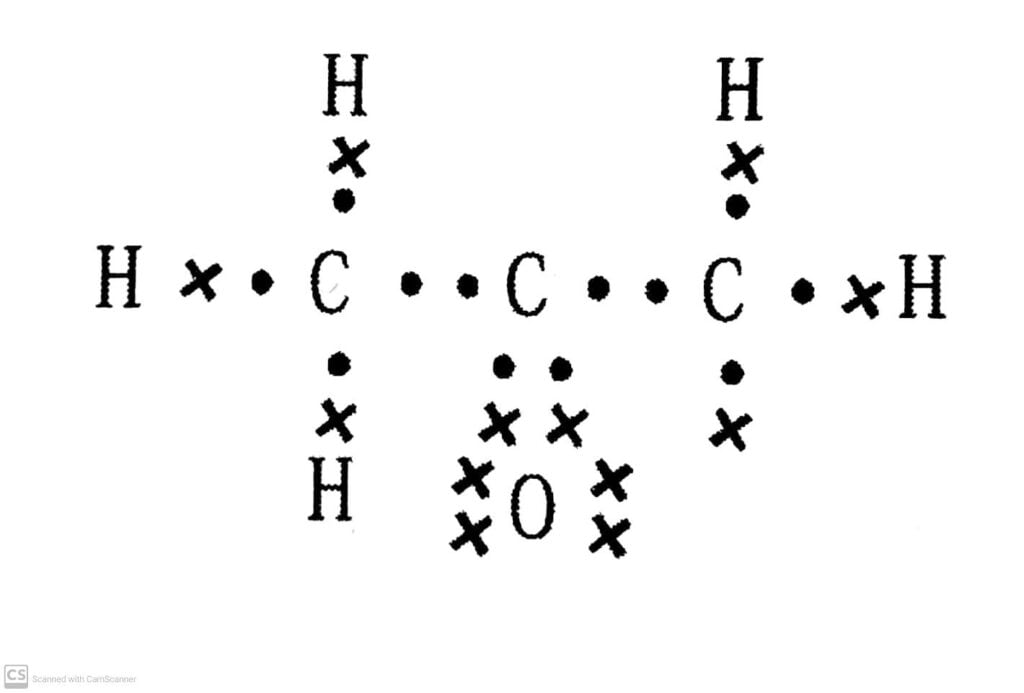
(d) F₂

Q.6. What is a homologous series? Explain with an example.
Ans: A homologous series is a series of carbon compounds that have different numbers of carbon atoms but contain the same functional group.For example methane ethane propane butane etc. are all part of the alkane homologous series.
The general formula of this series is CnH2n+2. Methane CH4 Ethane CH3CH3 Propane CH3CH2CH3 Butane CH3CH2CH2CH3 It can be noticed that there is a difference of −CH2 unit between each successive compound.
Q.7. How can ethanol and ethanoic acid be differentiated on the basics of their physical and chemical properties.
Ans: (i) Ethanol has a pleasant smell whereas ethanoic acid has the smell of vinegar.
(ii) Ethanol has a burning tase whereas ethanoic acid has a sour taste.
(iii) Ethanol has no action on litmus paper whereas ethanoic acid turns blue litmus paper to red.
(iv) Ethanol has no reaction with sodium hydrogen carbonate but ethanoic acid gives brisk effervescence with sodium hydrogen carbonate.
Q.8. Why does micelle formation take place when soap add to water? Will a micelle be formed in other solvents such as ethanol also?
Ans: Micelle formation takes place when soap is added to water because the hydrocarbon chains of soap molecules are hydrophobic which are insoluble in water, but the ionic ends of soap molecules are hydrophilic and hen soluble in water.
Micelle formation not take place when soap is added to organic solvents like ethanol because the hydrocarbon chains of soap molecules are soluble in organic solvents like ethanol.
Q.9. Why are carbon and its compounds used as fuels for most applications?
Ans: Carbon and its compounds used as fuels for most applications because they gives a large amount of heat per unit weight.
Q.10. Explain the formation of scum when hard water is treated with soap.
Ans: Hard water contains salts of calcium and magnesium. Calcium and magnesium on reaction with soap forms insoluble substance kcalled scun.
Q.11. What change will year observe if you test soap with litmus paper (red or blue)?
Ans: Red litmus paper turn blue.
Q.12. What is hydrogenation? What is its industrial application?
Ans: The addition of hydrogen to unsaturated hydrocarbon in the presence of catalyst is called hydrogenation.
Ghee on industrial scale is made by hydrogenation of naturally available vegetable oils.
Q.13. Which of the following hydrocarbons undergo addition reactions: C₂H₆, C₃H₈, C₃H₆, C₂H₂
Ans: C₃H₆ and C₂H₆
Q.14. Give a test that can be used to differentiate chemically between butter and cooking oil.
Ans: When a drop of bromine is added to cooking oil, its colour disappears whereas when a drop of bromine is added to butter, it becomes brown.
Q.15. Explain the mechanism of the cleaning action of soap.
Ans: When soap is dissolved in water, it forms a colloidal suspension in water in which the soap molecules cluster together to form spherical micelles. In a soap micelle, the soap molecules are arranged radially with hydrocarbon ends directed towards the centre and ionic ends directed out wards.
Multiple choice questions:
Q.1. Buckminsterfullerene is an allotropic form of the element:
(a) Phosphorus.
(b) Fluorine.
(c) Carbon.
(d) Sulphur.
Ans: (C) Carbon.
Q.2. Out of the following pairs of compounds, the unsaturated compounds are:
(a) C₂H₆ and C₄H₆
(b) C₆H₁₂ and C₅H₁₂
(c) C₄H₆ and C₆H₁₂
(d) C₂H₆ and C₄H₁₀
Ans: (c) C₄H₆ and C₆H₁₂
Q.3. The number of covalent bonds in pentane is:
(a) 5
(b) 12
(c) 17
(d) 16
Ans: (d) 16
Q.4. The properly of self – combination of the atoms of the same element to form long chains is known as:
(a) Protonation.
(b) Carbonation.
(c) Coronation.
(d) Catenation.
Ans: (d) Catenation.
Q.5. A cyclic hydrocarbon having carbon – carbon single bonds as well as carbon – carbon double bonds in its molecule is
(a) C₆H₁₂
(b) C₆H₁₄
(c) C₆H₆
(d) C₆H₁₀
Ans: (c) C₆H₆
Q.6. The hydrocarbon 2 – methylbutane is an isomer of
(a) N – pentane.
(b) N – butanes.
(c) Propane.
(d) Iso – butane.
Ans: (a) N – pentane.
Q.7. An unsaturated hydrocarbon having a triple co – valent bond has 50 hydrogen atoms in its molecule . The number of carbon atoms in its molecule will be
(a) 24
(b) 25
(c) 26
(d) 28
Ans: (c) 26
Q.8. An alkyne has seventy five carbon atoms in its molecule. The number of hydrogen atoms in its molecule will be
(a) 150
(b) 148
(c) 152
(d) 146
Ans: (b) 148
Q.9. A diamond toothed saw is usually used for cutting
(a) Stell girders.
(b) Logs of wood.
(c) Marble slabs.
(d) Asbestos sheets.
Ans: (c) Marble slabs.
Q.10. The organic compound prepared by Worker from an inorganic compound called ammonium cyanate was
(a) Glucose.
(b) Urea.
(c) Uric acid.
(d) Vinegar.
Ans: (b) Urea.
Q.11. One of the following is not an allotrope of carbon . This is –
(a) Diamond.
(b) Graphite.
(c) Cumene.
(d) Buckminsterfullerene.
Ans: (c) Cumene.
Q.12. The number of carbon atoms in the organic compound named as 2, 2, dimethylpropane is:
(a) Two.
(b) Five.
(c) Three.
(d) Four.
Ans: (b) Five.
Q.13. The pair of elements which exhibits the property of catenation is
(a) Sodium and silicon.
(b) Chlorine and carbon.
(c) Carbon and sodium.
(d) Silicon and carbon.
Ans: (d) Silicon and carbon.
Q.14. A saturated hydrocarbon has fifty hydrogen atoms in its molecule. The number of carbon atoms in its molecule will be:
(a) 25
(b) 24
(c) 26
(d) 27
Ans: (b) 24.
Q.15. A hydrocarbon having one double bond has 100 carbon atoms in its molecule. The number of hydrogen atoms in its molecule will be:
(a) 200
(b) 198
(c) 202
(d) 196
Ans: (a) 200.
Q.16. The hydrocarbon which has alternate single and double 2 bonds arranged in the form of a ring is:
(a) Cyclobutane.
(b) Benzene.
(c) Butene.
(d) Hexene.
Ans: (b) Benzene.
Q.17. Which of the following can not exhibit isomerism?
(a) C₄H₁₀
(b) C₅H₁₂
(c) C₃H₈
(d) C₆H₁₄
Ans: (c) C₃H₈
Q.18. The pencil leads are mainly made of:
(a) Lithium.
(b) Charcoal.
(c) Lead.
(d) Graphite.
Ans: (d) Graphite.
Q.19. The number of isomers formed by the hydrocarbon with molecular formula C₅H₁₂ is
(a) 2
(b) 5
(c) 3
(d) 4
Ans: (c) 3.
Q.20. The number of carbon atoms joined in a spherical molecules of buckminsterfullerene is –
(a) 50
(b) 60
(c) 70
(d) 90
Ans: (b) 60.
Q.21. The molecular formula of a homologue of butane is
(a) C₄H₈
(b) C₂H₆
(c) C₄H₆
(d) C₃H₈
Ans: (d) C₃H₈
Q.22. One of the following molecular formula can represents two organic compounds having different functional groups. This molecular formula is:
(a) C₅H₁₂O
(b) C₅H₁₀O
(c) C₅H₁₀O₂
(d) C₅ H₁₂
Ans: (b) C₅H₁₀O
Q.23. The number of carbon atoms present in the molecule of fifth member of the homologous series of alkynes is
(a) Four.
(b) Five.
(c) Six.
(d) Seven.
Ans: (c) Six.
Q.24. One of the following burns without producing flame. This is:
(a) Wood.
(b) Charcoal.
(c) LPG.
(d) Candle.
Ans: (b) Charcoal.
Q.25. The functional group which always occurs in the middle of a carbon chain as:
(a) Alcohol.
(b) Aldehyde group.
(c) Carboxyl group.
(d) ketone group.
Ans: (d) ketone group.
Q.26. The molecular formula of some organic compounds are given below. Which of these compounds contains an aldehyde group?
(a) C₃H₈O
(b) C₃H₆O₂
(C) C₃H₆O
(d) C₃H₇CI
Ans: (c) C₃H₆O
Q.27. The organic compounds which are isomeric with one another are:
(a) Alcohols and aldehydes.
(b) Aldehydes and carboxylic acids.
(c) ketenes and aldehydes.
(d) Alcohols and ketenes.
Ans: (c) Ketones and aldehydes.
Q.28. The fuel which usually burns with a blue flame is
(a) Coal.
(b) LPG.
(c) Candle wax.
(d) kerosene (in lamp).
Ans: (b) LPG.
Q.29. Which of the following burns by producing a yellow, luminous flame?
(a) Natural gas.
(b) Coke.
(c) Wax.
(d) Charcoal.
Ans: (c) Wax.
Q.30. The molecular formula of an organic compound is C₄₈H₉₄. This compound belongs to the homologous series of:
(a) Alkenes.
(b) Aldehydes.
(c) Alkynes.
(d) Alkaness.
Ans: Alkynes.
Q.31. One of the following molecular formule represents as ketone. This formula is:
(a) C₅H₁₂O
(b) C₆H₁₂O
(c) C₆H₁₄O
(d) C₆H₁₂O
Ans: (d) C₆H₁₂O
Q.32. Which one of the following in not a fossil fuel?
(a) Petrol.
(b) Coal.
(c) Charcoal.
(d) Coal.
Ans: (c) Charcoal.
Q.33. Butanone is a four carbon compound having the functional group:
(a) -COOH
(b) -CHO
(c) -CO-
(d) -OH
Ans: (c) -CO-
Q.34. The molecular formula of the third member of the homologous series of ketones is:
(a) C₄H₈O
(b) C₃H₆O
(c) C₅H₁₀O
(d) C₆H₁₂O
Ans: (c) C₅H₁₀O
Q.35. The functional group present in propanal is:
(a) -OH
(b) -COOH
(c) -CO-
(d) -CHO
Ans: (d) -CHO
Q.36. While cooking , if the bottom of the utensil is getting blackned on the outside, it means that:
(a) The food is not cooked completely.
(b) The fuel is not burning completely.
(c) The fuel is wet U
(d) The fuel is burned completely .
Ans: (b) The fuel is not burning completely.
Q.37. When ethanol is heated with alkaline potassium permanganate solution, it gets converted into ethanoic acid. In this reaction alkaline potassium permanganate bay act as:
(a) Reducing agent.
(b) Oxidising agent.
(c) Catalyst sa ozbr.
(d) Dehydrating agent.
Ans: (b) Oxidising agent.
Q.38. When ethanol is heated with concentrated sulphuric acid at 170° C, it gets converted into ethene. In this reaction concentrated sulphuric acid acts as:
(a) Oxidising agent.
(b) Catalyst.
(c) Dehydrating agent.
(d) Reducing agent.
Ans: (c) Dehydrating agent .
Q.39. When a vegetable oil is treated with hydrogen in the presence of nickel catalyst, it forms a fat. This is an example of:
(a) Anodising reaction.
(b) Substitution reactions.
(c) Displacement reaction.
(d) Addition reaction.
Ans: (d) Addition reaction.
Q.40. The soap molecule has a:
(a) Hydrophilic head and a hydrophobic tail.
(b) Hydrophobic head and hydrophilic tail.
(c) Hydrophobic head and hydrophobic tail
(d) Hydrophilic head and hydrophilic tail .
Ans: (a) Hydrophilic head and a hydrophobic tail.
Q.41. Chlorine reacts with saturated hydrocarbons at room temperature in the:
(a) Absence of sunlight.
(b) presence of sunlight.
(c) Absence of moisture.
(d) Presence of H₂SO₄
Ans: (b) Presence of sunlight.
Q.42. In a soap micelle, the soap molecules are arranged radially with:
(a) Ionic ends directed towards the centre and hydrocarbon ends directed out wards.
(b) Hydrocarbon ends directed towards the centre and ionic ends directed outwards.
(c) Both ionic ends and hydrocarbon ends directed towards the centre.
(d) both hydrocarbon ends and ionic ends directed outwards.
Ans: (b) Hydrocarbon ends directed towards the centre and ionic ends directed outwards.
Q.43. When ethanol reacts with sodium metal, it forms two products. These products are:
(a) Sodium ethanoate and oxygen.
(b) Sodium ethanoate and hydrogen.
(c) Sodium ethoxide and oxygen.
(d) Sodium ethoxide and hydrogen.
Ans: (d) Sodium ethoxide and hydrogen.
Q.44. Vinegar is a solution of about
(a) 5 to 8 percent ethanoic acid in alcohol.
(b) 5 to 8 percent ethanoic acid in water.
(c) 50 to 80 percent ethanoic acid in water.
(d) 50 to 80 percent ethanoic acid in alcohol.
Ans: (b) 5 to 80 percent ethanoic acid in water .
Q.45. One of the following substance is not added to make denatured alcohol. This is
(a) Methyl alcohol.
(b) Copper sulphate.
(c) Chloroform.
(d) Pyridine dam.
Ans: (c) Chloroform.
Q.46. One of the following organic compounds cannot decolourise the red – brown colour of bromine water . This compound is
(a) C₁₄H₂₈
(b) C₇H₁₂
(c) C₆H₁₄
(d) C₉H₁₆
Ans: (c) C₆H₁₄
Q.47. The substance which can produce brisk effervescence with baking soda solution is –
(a) Ethanol.
(b) Vegetable oil.
(c) Vinegar.
(d) Common Salt.
Ans: (c) Vinegar.
Q.48. The chemical which is not required for the preparation of soap in the laboratory is:
(a) Vegetable oil.
(b) Baking soda.
(c) Caustic soda.
(d) Common salt.
Ans: (b) Baking soda.
Q.49. Which of the following can damage optic nerve leading to blindness, if taken internally?
(a) CH₃ COOH
(b) C₂H₅OH
(c) NaHCO₃
(d) CH₃ OH
Ans: (d) CH₃ OH
Q.50. The usual disease caused by the excessive drinking alcohol over a long period of time is:
(a) Diabetes.
(b) Cataract.
(c) Cirrhosis.
(d) Arthritis.
Ans: (c) Cirrhosis.
Q.51. Which of the following molecular formula corresponds to ethyl butanoate ester?
(a) C₅H₁₀O₂
(b) C₆H₁₂O₂
(c) C₇H₁₄O₂
(d) C₈H₁₈O₂
Ans. (b) C₆H₁₂O₂

Hi, I’m Dev Kirtonia, Founder & CEO of Dev Library. A website that provides all SCERT, NCERT 3 to 12, and BA, B.com, B.Sc, and Computer Science with Post Graduate Notes & Suggestions, Novel, eBooks, Biography, Quotes, Study Materials, and more.

