NIOS Class 12 Chemistry Chapter 9 Chemical Thermodynamics Solutions English Medium As Per New Syllabus to each chapter is provided in the list so that you can easily browse throughout different chapters NIOS Class 12 Chemistry Chapter 9 Chemical Thermodynamics Notes in English and select need one. NIOS Class 12 Chemistry Solutions English Medium Download PDF. NIOS Study Material of Class 12 Chemistry Notes Paper Code: 313.
NIOS Class 12 Chemistry Chapter 9 Chemical Thermodynamics
Also, you can read the NIOS book online in these sections Solutions by Expert Teachers as per National Institute of Open Schooling (NIOS) Book guidelines. These solutions are part of NIOS All Subject Solutions. Here we have given NIOS Class 12 Chemistry Notes, NIOS Senior Secondary Course Chemistry Solutions in English for All Chapter, You can practice these here.
Chemical Thermodynamics
Chapter: 9
| Module – IV: Chemical Energetics |
INTEXT QUESTIONS 9.1
1. Which of the following is false?
(a) The reaction.
H₂(g) + Cl₂(g) → 2HCl (g) + 185 kJ is endothermic.
(b) Enthalpy change is a state function.
(c) Standard state condition for a gaseous system is 1 bar pressure at a specified temperature.
Ans: (a).
2. For the reaction at 298 K,
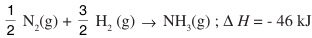
(a) What is the value of Δng?
(b) Calculate the value of Δ U at 298 K?
Ans: (a) Δn = 1 – (1)/(2) – (3)/(2) = – 1
(b) ΔU = ΔH – ΔnRT
= 46000 (J mol⁻¹) – (– 1) (8.314 Jk–1 mol–1) × (298 K)
= – 46000 (J mol⁻¹) + 2247.6 (J mol–1)
= – 43.5 kJ mol⁻¹
3. Which of the following will increase the internal energy of a system?
(a) Heat given to the system.
(b) Work done by the system.
Ans: (a) Heat given to the system.
INTEXT QUESTIONS 9.2
1. Which of the following is true?
(a) Enthalpy of formation is the heat evolved or absorbed when one gram
of a substance is formed from its elements in their most stable states.
(b) When one mole of H+ (aq) and 1 mole of OH— (aq) react, 57.1 kJ of energy is absorbed.
(c) In the thermochemical equation, C(graphite) + O₂ (g) → CO₂(g) ; ΔfH⁰ = –394 kJ mol–1
Δ fH⁰ is known as enthalpy of formation of CO₂(g)
Ans: (c).
2. Calculate the enthalpy change for complete combustion of 29.0 g of butane, if:
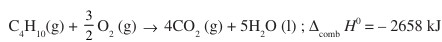
Ans: – 1329 kJ
3. Calculate the standard enthalpy of the reaction:
2H₂S(g) + SO₂(g) → 3S (s) + 2H₂O (l)given that:
Δf H⁰ (H₂S) = – 20.6 kJ mol⁻¹
Δf H⁰ (SO₂) = – 296.9 kJ mol⁻¹
Δf H⁰ (H₂O) = – 289.9 kJ mol⁻¹
Ans: 3Δr H⁰ = 2 Δf H⁰ (H₂O) – 2ΔfH⁰
(H2₂S) – ΔfH⁰ (SO₂) = – 241.7 kJ
INTEXT QUESTIONS 9.3
1. Write True or false:
(a) Enthalpy of a reaction is equal to the sum of the enthalpy of formation of products minus the sum of the enthalpy of formation of the reactants.
Ans: True.
(b) Enthalpy of formation of any elementary substance is equal to zero.
Ans: True.
(c) If a reaction can be carried out in several steps, the enthalpy change for the overall reaction is equal to enthalpy change in the last step.
Ans: False.
(d) Bond enthalpy and bond dissociation energy are same for polyatomic molecules.
Ans: False.
2. Calculate the bond enthalpy of N – H in NH₃ (g), given
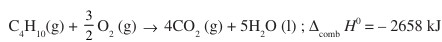
1/2 H2 (g) → H(g) Δf Ho = 218 kJ mol-1
1/2 N2 (g) → N(g) ΔfHo = 973 kJ mol-1
Ans: ΔrH⁰ = ∑ B.E. (reactant bonds) – ∑ B.E. (products bonds) or – 46 (kJ mol–1) = 3 × 218 (kJ mol⁻¹) + 973 (kJ mol⁻¹) – B.E. (NH₃(g)Bonds)
B.E. (NH₃(g) Bonds) = 1673 kJ mol⁻¹
B.E. (N – H) = 557.7 kJmol⁻¹
3. Calculate the enthalpy of the reaction:
H₂ (g) + Cl₂ (g) → 2HCl (g)
given;
Bond enthalpy (H – H) = 435 kJ mol⁻¹
Bond enthalpy (Cl – Cl) = 242 kJ mol⁻¹
Bond enthalpy (H – Cl) = 431 kJ mol⁻¹
Ans: Δr H⁰ = – 185 k mol⁻¹

Hi! my Name is Parimal Roy. I have completed my Bachelor’s degree in Philosophy (B.A.) from Silapathar General College. Currently, I am working as an HR Manager at Dev Library. It is a website that provides study materials for students from Class 3 to 12, including SCERT and NCERT notes. It also offers resources for BA, B.Com, B.Sc, and Computer Science, along with postgraduate notes. Besides study materials, the website has novels, eBooks, health and finance articles, biographies, quotes, and more.


 Buy Now
Buy Now