Class 11 Physics Important Chapter 13 Kinetic Theory Solutions English Medium As Per AHSEC New Syllabus to each chapter is provided in the list so that you can easily browse through different chapters NCERT Class 11 Physics Important Chapter 13 Kinetic Theory and select need one. AHSEC Class 11 Physics Additional Notes English Medium Download PDF. HS 1st Year Physics Important Solutions in English.
Class 11 Physics Important Chapter 13 Kinetic Theory
Also, you can read the NCERT book online in these sections Solutions by Expert Teachers as per Central Board of Secondary Education (CBSE) Book guidelines. NCERT Class 11 Physics Additional Question Answer are part of All Subject Solutions. Here we have given HS 1st Year Physics Important Notes in English for All Chapters, You can practice these here.
Kinetic Theory
Chapter: 13
| IMPORTANT QUESTION AND ANSWER |
Answer the Following Questions:
1. What is the Kinetic Theory of Gases?
Ans: The Kinetic Theory of Gases explains the behavior of gases in terms of the motion of their constituent molecules. It assumes that gas molecules are in continuous random motion, and their collisions with the walls of the container produce pressure. The theory is fundamental in understanding various gas laws and molecular properties, including temperature and pressure.
2. State and explain Boyle’s Law.
Ans: Boyle’s Law states that for a given amount of gas at constant temperature, the pressure of the gas is inversely proportional to its volume.
P ∝ 1/V
Where P is pressure and V is volume. This means that if the volume of the gas decreases, the pressure increases, provided the temperature remains constant.
3. What is Avogadro’s Law?
Ans: Avogadro’s Law states that equal volumes of all gases at the same temperature and pressure contain an equal number of molecules. This law was instrumental in establishing the concept of the mole and the atomic hypothesis.
V ∝ N
Where V is the volume and N is the number of molecules.
4. Explain the law of equipartition of energy.
Ans: The law of equipartition of energy states that at thermal equilibrium, the total energy of a system is equally distributed among its different degrees of freedom. Each degree of freedom contributes an energy of 1/2kBT, where kB is the Boltzmann constant and T is the temperature in Kelvin.
(i) Translational degrees of freedom: 1/2kBT
(ii) Rotational degrees of freedom: 1/2kBT
(iii) Vibrational degrees of freedom: kBT for each mode (kinetic and potential energy).
5. What is the relationship between temperature and kinetic energy according to the Kinetic Theory?
Ans: According to the Kinetic Theory of Gases, the average kinetic energy of gas molecules is directly proportional to the absolute temperature of the gas.
E = 3/2kBT
Where E is the average kinetic energy and T is the absolute temperature.
6. Describe the concept of an ideal gas.
Ans: An ideal gas is a hypothetical gas that perfectly obeys the ideal gas equation:
PV = nRT
Where P is the pressure, V is the volume, nnn is the number of moles, R is the gas constant, and T is the temperature in Kelvin. Real gases deviate from ideal behavior, especially at high pressures and low temperatures.
7. What is the significance of the Boltzmann constant kB?
Ans: The Boltzmann constant kB relates the average kinetic energy of a particle in a gas to the temperature of the gas. It serves as a bridge between macroscopic and microscopic physical quantities, linking temperature to energy.
8. Explain Dalton’s Law of Partial Pressures.
Ans: Dalton’s Law of Partial Pressures states that in a mixture of non-reacting gases, the total pressure exerted by the gas mixture is the sum of the partial pressures exerted by each individual gas.
Ptotal = P1+P2+⋯+Pn
Where P1,P2,…are the partial pressures of individual gases.
9. What is the concept of ideal gas law, and how does it relate to kinetic theory?
Ans: The ideal gas law is a relation between pressure, volume, temperature, and the number of moles of a gas, given by the equation:
PV = nRT
According to the kinetic theory, gases are made up of molecules in constant random motion. The pressure exerted by the gas is a result of these molecules colliding with the walls of the container. The ideal gas law relates macroscopic properties (pressure, volume, temperature) to microscopic properties (molecular motion, molecular interactions).
10. State and explain Charles’ Law.
Ans: Charles’ Law states that for a given mass of gas at constant pressure, the volume of the gas is directly proportional to its absolute temperature.
V∝T
Where V is the volume and T is the temperature. If the temperature of a gas increases, the volume also increases, provided the pressure is constant. This relationship is a direct consequence of the kinetic theory, as increasing temperature increases the average kinetic energy of the molecules, causing them to move faster and occupy more space.
11. How does the kinetic theory explain the pressure exerted by a gas?
Ans: The kinetic theory explains that gas pressure is caused by the continuous collisions of gas molecules with the walls of the container. The force exerted during each collision contributes to the overall pressure. The pressure PPP of the gas is related to the number density of molecules nnn, the average kinetic energy of molecules, and the temperature of the gas as:
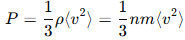
Where mmm is the mass of a molecule and v2 is the average squared velocity of the molecules.
12. What is the difference between real gases and ideal gases?
Ans: Ideal gases are hypothetical gases that perfectly follow the ideal gas law and exhibit no intermolecular forces, meaning that they are free to move in straight lines and collide elastically. Real gases, however, deviate from ideal behavior, especially at high pressures and low temperatures. Real gases experience intermolecular attractions and repulsions, which affect their behavior, especially at high densities and low temperatures. The Van der Waals equation is often used to describe the behavior of real gases:
(P + a/V2)(V−b) = RT
Where a and b are constants that account for intermolecular forces and molecular size.

Hi! my Name is Parimal Roy. I have completed my Bachelor’s degree in Philosophy (B.A.) from Silapathar General College. Currently, I am working as an HR Manager at Dev Library. It is a website that provides study materials for students from Class 3 to 12, including SCERT and NCERT notes. It also offers resources for BA, B.Com, B.Sc, and Computer Science, along with postgraduate notes. Besides study materials, the website has novels, eBooks, health and finance articles, biographies, quotes, and more.




 Buy Now
Buy Now