Class 10 Science Chapter 2 Acids-Bases and Salts The answer to each chapter is provided in the list so that you can easily browse throughout different chapters NCERT Class 10 Science Chapter 2 Acids-Bases and Salts and select need one.
Class 10 Science Chapter 2 Acids-Bases and Salts
Also, you can read the SCERT book online in these sections Solutions by Expert Teachers as per SCERT (CBSE) Book guidelines. These solutions are part of SCERT All Subject Solutions. Here we have given Assam Board Class 10 Science Chapter 2 Acids-Bases and Salts Solutions for All Subjects, You can practice these here.
Acids-Bases and Salts
Chapter – 2
GENERAL SCIENCE
Textual Questions and Answers:
Page – 18
Q.1. You have been provided with three test tubes. One of them contains distilled water and the other two contain an acidic solution and a basic solution, respectively. If you are given only red litmus paper. How will you identify the contents of each test tube?
Ans: Using red litmus paper, we can determine the contents of each test tube.
This can be accomplished by observing the color shift of the red litmus paper:
- A few drops of each of the 3 samples in the test tubes are added separately to the litmus paper.
- A basic solution in one of the test tubes turns red litmus paper blue.
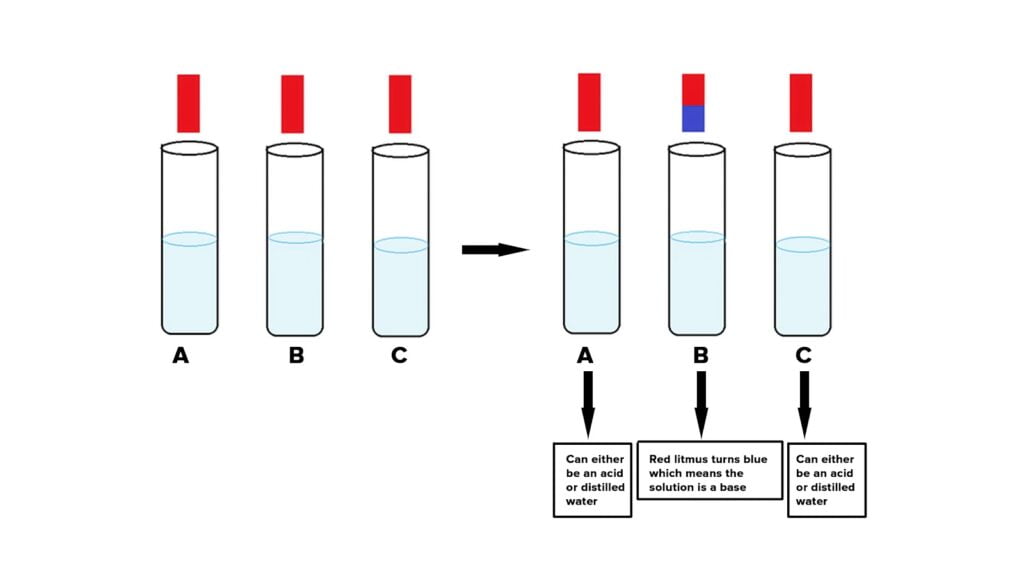
- Separate the blue litmus paper into two halves and a few drops of each of the remaining 2 samples in the test tubes should be added separately to the blue litmus paper.
- Now, the acidic solution will be the solution in the test tube that turns blue litmus paper red.
- Distilled water is present in test-tube solutions that do not change the color of either red or blue litmus paper.
Page No – 22
Q.1. Why should curd and sour substances not be kept in a brass and copper vessels?
Ans: Brass and copper are metals.
- Curd and other sour substances contain lactic acid (C 3 H 6 O 3 ).
- When metals react with acids , they produce salt and hydrogen gas.
- When sour substances like curd are kept in brass or copper vessels , the lactic acid in curd reacts with the metals which leads to the corrosion of the vessels as well as the formation of harmful salts that cannot be consumed.
Q.2. Which gas is usually liberated when an acid react with a metal. Illustrate with an example. How will you test for the presence of the gas?
Ans: (i) H₂ gas is liberated when an acid reacts with a metal.
(ii) Illustration: Set up an apparatus. Take some Zinc granules in the test tube. Add about 5 ml dilute hydrochloric acid slowly. Soon the reaction between Zinc and hydrochloric acid starts and hydrogen gas is evolved.
(iii) Test for H₂ gas: H₂ gas is not soluble in water. When passed through soap solution, it gets trapped into bubbles which burn with explosion.
Q.3. Metal compound a reacts with dilute hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle. Write a balanced chemical equation for the reaction if one of the compounds formed is calcium chloride.
Ans: Since the end product is calcium chloride and the gas formed extinguishes a burning candle, it is Co₂ . The metal compound must be calcium carbonate.
Hence, the reaction between calcium carbonate and hydrochloric acid is as follows:
C aCO₃ + 2HCI → CaCl₂ + CO₂ + H₂O
Page – 25
Q.1. Why do HCI, HNO₃ etc., show acidic characters in aqueous solutions while solutions of compounds like alcohol and glucose do not show acidic character?
Ans: Solutions like HCl, HNO3 etc. get ionised in aqueous solutions and due to the presence of H⁺ ions they show acidic characters. While solutions of compounds like alcohol and glucose do not form any such ions so they do not show acidic characters.
Q.2. Why does an aqueous solution of an acid conduct electricity?
Ans: In the liquid state, pure acids are terrible conductors of electricity. The presence of ions in aqueous solutions of acids is demonstrated by the fact that they conduct electricity. In an aqueous solution, acids dissociate to create H⁺ ions.
H⁺ ions reach the cathode when electricity is transmitted through an aqueous solution of an acid, and each H⁺ ion picks up one electron from the cathode to generate H₂ gas. An aqueous acid solution conducts electricity as a result of this reaction.
HCl(aq) → H⁺(aq) + Cl⁻(aq)
In an aqueous solution with water as the solvent, the solute to be dissolved by the solvent contains fewer particles, causing the particles to move randomly. Simply said, the aqueous solution of acid conducts electricity due to the presence of ions.
Q.3. Why does dry Hcl gas not change the colour of the dry litmus paper?
Ans: The colour of litmus paper is changed by the hydrogen ions. Dry HCl does not contain hydrogen ions (H+). Acid (For example; HCl) dissociates only in aqueous solution to give ions. since, in this case, neither HCl is in the aqueous form nor litmus paper is wet.
Therefore, the colour of litmus paper does not change.
Q.4. While diluting au acid, why is it recommended that the acid should be added to water and not water to the acid?
Ans: The acid must always be added slowly to water with constant stirring. If water is added to a concentrated acid, the heat generated may cause the mixture to splash out and cause burns. The glass container may also break due to excessive local heating.
Q.5. How is the concentration of hydronium ions (H₃O⁺) affected when a solution of an acid is diluted?
Ans: Mixing an acid with water results in decrease in the concentration of H₃O⁺ ions per unit volume.
Q.6. How is the concentration of hydroxide ions (OH⁻) affected when excess base is dissolved in a solution of sodium hydroxide?
Ans: The concentration of hydroxide ions will increase when excess base is dissolved in a solution of sodium hydroxide because the amount of hydroxide ions per unit volume increases. This happens only when base added dissolved in water. If the base is not soluble in water, the concentration of hydroxide ions remains constant.
Page – 28
Q.1. You have two solutions, A and B. The Pᴴ of solution A is 6 and Pᴴ of solution B is 8. Which solution has more hydrogen ion concentration? Which of this is acidic and which one is basic?
Ans: A solution of pH value less than 7 is acidic in nature, while greater than 7 indicates a basic solution. Therefore, the solution with pH = 6 is acidic and has more hydrogen ion concentration than the solution of pH = 8, which is basic.
Q.2. What effect does the concentration of H⁺ (aq) ions have on the nature of the solution?
Ans: Acids that give rise to more H⁺ ions are strong acids and acids that give less H⁺ ions are weak acids.
Q.3. Do basic solutions also have H⁺ (aq) ions ? If yes, then why are these basic?
Ans: Yes, H+ ions can be found in all basic solutions. But these are farless in number than OH⁻ ions. For more number of OH⁻ ions the solution is basic.
Q.4. Under what soil condition do you think a farmer would treat the soil of his fields with quick lime (calcium oxide) or slaked lime (calcium hydroxide) or chalk (calcium carbonate)?
Ans: If the soil is acidic, it affects the growth of cultivation. When the soil become acidic farmers treat the soil fields with bases like quick lime, slaked lime or chalk.
Page – 33
Q.1. What is the common name of the compound CaOcl₂?
Ans: Bleaching powder.
Q.2. Name the substance which on treatment with chlorine yields bleaching powder.
Ans: Calcium Hydroxide.
Q.3. Name the sodium compound which is used for softening hard water.
Ans: Sodium Carbonate.
Q.4. What will happen if a solution of sodium hydrocarbonate is heated? Give the equation of the reaction involved.
Ans: When sodium hydrogen carbonate is heated, sodium carbonate and water are formed along with the evolution of carbon dioxide gas.
The reaction involved is: 2NaHCO₃ → Na₂Co₃ + Co₂+ H₂O
Q.5. Write an equation to show the reaction between plaster of paris and water.
Ans:
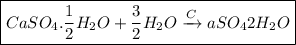
Plaster of Paris when reacts with water liberating heat through crystallization. Gypsum is produced by heating plaster of paris to about 150-degree Celsius. When the dry plaster of paris powder is mixed with water it reforms into gypsum. Depending upon the temperature gypsum converts to hemihydrate or anhydrous form.
EXERCISES
Q.1. A solution turns red litmus blue, its Pᴴ is likely to be
(a) 1
(b) 4
(c) 5
(d) 10
Ans: (d) 10.
Q.2. A solution reacts with crushed egg- shells to give a gas that turns lime water milk. The solution contains.
(a) Nacl
(b) Hcl
(c) Licl
(d) Kcl
Ans: (b) Hcl.
Q.10 ml of solution of NaOH is found to be completely neutralised by 8 ml of a given solution of Hcl. If we take 20 ml of the same solution of NaOH, the amount Hcl solution (the solution as before) required to neutralise it will be
(a) 4 ml
(b) 8 ml
(c) 12 ml
(d) 16 ml
Ans: (d) 16 ml.
Q.4. Which one of the following types of medicines is used for treating indigestion?
(a) antibiotic.
(b) Analgesic.
(c) Antacid.
(d) Antiseptic.
Ans: (c) Antacid.
Q.5. Write word equations and then balanced equations for the reaction taking place when:
(a) dilute sulphuric acid reacts with zinc granules.
(b) dilute hydrochloric acid reacts with magnesium.
(c) dilute sulphuric acid reacts with aluminium powder.
(d) dilute hydrochloric acid reacts with iron filings.
Ans: (a) Dil Sulphuric acid + zinc → zinc sulphate + Hydrogen H₂SO₄ + zn ZnSO₄ + H₂
(b) Dil hydrochloric acid + Magnesium → Magnesium chloride + Hydrogen.
Hcl + Mg → Mgcl₂ + H₂
⇒ 2HCl + Mg → Mgcl₂ + H₂
(c) Aluminium powder + dil sulphuric acid → Aluminium sulphate + Hydrogen Al + H₂SO₄ → Al₂(SO₄)₃ + H₂
⇒ 2AI + 3H₂SO₄ → Al₂ (SO₄)₃ + H₂
(d) Dil. hydrochloric acid + Iron → Iron chloride + Hydrogen Hcl + Fe → FeCl₃ + H₂
⇒ 6Hcl + 2Fe → 2Fecl₃ +3H₂
Q.6. Compounds such as alcohols and glucose also contain hydrogen beet are not categorised as acids. Describe an activity to prove it.
Ans: Take solutions of glucose and alcohols in a beaker. Fix two nails on a cork and place the cork in the beaker. connect the nails to the two terminals of a 6 volt battery through a bulb and a switch. Switch on the current. The bulb does not glow. That means current does not pass through the circuit. This show that no H⁺ ions are present in the solution. This experiment shows that alcohol and glucose are not acid.
Q.7. Why does distilled water not conduct electricity, whereas rain water does?
Ans: Distilled water is pure form of water which do not contain any solute in it. Therefore it cannot conduct electricity because it does not contain ions while rain water contains dissolved salts and acids which dissociates in ions and conducts electricity.
Q.8. Why do acids not show acidic behaviour in the absence of water?
Ans: Acids form hydrogen ions [H+ (aq) ions] in its aqueous solution which are responsible for their acidic characters. Therefore acids cannot show acidic behaviour in the absence of water.
Q.9. Five solutions A, B, C, D and E when tested with universal indicator showed Pᴴ as 4, 1, 11, 7 and 9 respectively. Which solution is
(a) Neutral?
(b) Strongly alkaline?
(c) Strongly acidic?
(d) Weakly acidic?
(e) Weakly alkaline?
Arrange the Pᴴ in increasing order of hydrogen ion concentration.
Ans: (a) Neutral solution is D
(b) Strongly alkaline is C
(c) Strongly acidic is B
(d) Weakly acidic is A
(e) Weakly alkaline is E
Pᴴ values in increasing order of hydrogen ion concentration 11<9<7<4<1
Q.10. Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (Hcl) is added to test tube A. while acetic acid (CH₃ COOH) is added to test tube B. Amount and concentration taken for both the acids are same. In which test tube will be fizzing occur more vigorously and why?
Ans: Hydrochloric acid is stronger acid than acetic acid and reaction between magnesium ribbon and Hcl is faster than between magnesium ribbon and CH₃ COOH. So fizzing will occur more vigorously in test tube A. containing hydrochloric acid.
Q.11. Fresh milk has a pH of 6. How do you think the Pᴴ will change as it turns into curd? Explain your answer.
Ans: The Pᴴ value of fresh milk falls below 6 when it turns into curd due to the formation of lactic acid during the process.
Q.12. A milkman adds a very small amount of baking soda to fresh milk.
(a) Why does the shift the Pᴴ of the fresh milk from 6 to slightly alkaline?
(b) Why does this milk take a long time to set as curd?
Ans: (a) The milkman adds a little baking soda to fresh milk to make it slightly alkaline so that milk can be preserved for a longer time.
(b) The lactic acid is used for neutralise the base initially and when more lactic acid is formed then the milk sets as curd.
Q.13. Plaster of paris should be stored in a moisture-proof container. Explain why?
Ans: If plaster of paris will store in a moisture then it changes to gypsum giving a hard solid mass. Which does not have the required selting property.
Q.14. What is a neutralisation reaction? Give two example.
Ans: A neutralization reaction is when an acid and a base react to form H2O and salt and involves the combination of H ions and hydroxyl ions to generate H2O. The neutralization of a strong acid and strong base has a pH equal to 7.
Example: When Sodium hydroxide is added to hydrochloric acid. Sodium chloride and water are formed.
Examples: (i) Hcl + NaOH → Nacl + H₂O
(acid) (Base) (Salt) (Water)
Q.15. Give two important uses of washing soda and baking soda.
Ans:
| Uses of washing soda | Uses of baking soda |
| (i) Washing soda is used in glass, soap and paper paper industries. | (i) For making baking powder, which is a mixture of baking soda and a mild edible acid such as tartaric acid. |
| (ii) It is used for removing permanent hardness of water. | (ii) It is used in soda-acid fire extinguishers. |
Multiple choice questions:
1. Out of these which one gas is used in fire extinguishers.
(a) CO₂
(b) SO₂
(c) NO₂
(d) H₂S
Ans: (a) CO₂
2. Milk of magnesia has
(a) Acidic nature.
(b) Basic nature.
(c) Amphoteric nature.
(d) Neutral.
Ans: (a) Acidic nature.
3. Which of the following will give Pᴴ less than seven?
(a) Blood.
(b) Milk of magnesia.
(c) Sodium hydroxide.
(d) Gastric juice.
Ans: (d) Gastric juice.
4. Which of the following compounds is used to repair fractured bones?
(a) Na₂CO₃
(b) CaOcl₂
(c) CaSo₄½ H₂O
(d) CaSo₄.5H₂O
Ans: (c) CaSo₄½ H₂O
5. Tooth enamel contains.
(a) Calcium carbonate.
(b) Calcium sulphate.
(c) Calcium chloride.
(d) Calcium phosphate.
Ans: (d) Calcium phosphate.
6. Which one of the following types of medicine is used for treating indigestion?
(a) Antibiotic.
(b) Analgesic.
(c) Antacid.
(d) Antiseptic.
Ans: (c) Antacid.
7. The solution of sodium hydroxide is
(a) Acidic.
(b) Alkaline.
(c) Neutrall.
(d) Amphoteric.
Ans: (b) Alkaline.
8. Name the acid present in lemon juice.
(a) Acetic acid.
(b) Citric acid.
(c) Propanoic acid.
(d) Sodium carbonate.
Ans. (b) Citric acid.
9. The natural indicator among the following is
(a) phenolph thalein.
(b) methyl orange.
(c) methyl red.
(d) litmus.
Ans: (d) litmus.
10. Basic solutions contain:
(a) H⁺ ions.
(b) OH⁻ ions.
(c) Both H⁺ and OH⁻ ions.
(d) Na⁺ ions.
Ans: (c) Both H⁺ and OH⁻ ions.
11. What is corrosion?
Ans: Metal that has been extracted from its primary ore, has a natural tendency to revert to its natural state under the action of oxygen and water. This action is called corrosion, and the most common example is the rusting of steel.
12. What is the other name of calcium hydroxide?
Ans: Slaked lime.
13. What is rock salt? Give example.
Ans: Halite more commonly known as Rock salt is a mineral formed from sodium chloride. It’s chemical formula is NaCl and this also includes other variations of salt such as common salt and table salt.
Example: Common salt (Nacl)
14. Name two naturally occurring acids.
Ans: 1. Citric acid. and
2. Acetic acid.
15. Name a hydrogen containing compound which does not act as an acid.
Ans: Methane (CH₄)

Hi, I’m Dev Kirtonia, Founder & CEO of Dev Library. A website that provides all SCERT, NCERT 3 to 12, and BA, B.com, B.Sc, and Computer Science with Post Graduate Notes & Suggestions, Novel, eBooks, Biography, Quotes, Study Materials, and more.

